 Chapter 18
Chapter 18 
LEARNING AND MEMORY
| Definitions |
What is learning? According to Eric Kandel (2000) “Learning is the process by which we acquire knowledge about the world.” While this definition is erudite, it doesn’t help us much in knowing what to study. Another definition (Kimble, 1961), "Learning refers to a more or less permanent change in behavior which occurs as a result of practice," is a little better. It tells us that learning is more or less permanent; it won’t always be there, but often will. It also tells us that this is something that happens because we practice–repeat something over and over. A further definition says, “[Learning is] either a case of differential strengthening of one from a number of responses evoked by a situation of need, or the formation of receptor-effector connections de novo; the first occurs typically in simple selective learning and the second in conditioned reflex learning" (Hull, 1943). It is the strengthening of existing responses and the formation of new responses to existing stimuli that make this definition unique. So what is learning? It isn’t clear that we have an inclusive definition. It appears that learning is the strengthening of existing responses or formation of new responses to existing stimuli that occurs because of practice or repetition. How much practice? Sometimes a single practice session is sufficient as in avoidance of painful or noxious stimuli. Sometimes a lot of practice is necessary as in learning to drive a car.
What then is memory? Again according to Kandel (2000), ". . . memory is the process by which that knowledge of the world is encoded, stored, and later retrieved." By this definition, memory is not a thing; it’s a process. Interesting! In another definition, "Memory is a phase of learning . . . learning has three stages: 1. acquiring, wherein one masters a new activity . . . or memorizes verbal material . . . 2. retaining the new acquisition for a period of time; and 3. remembering, which enables one to reproduce the learned act or memorized material. In a narrower sense learning merely means acquiring skill . . ." (Sargent & Stafford, 1965). From these definitions, we see that memory has to do with keeping “knowledge” someplace and then retrieving it when it is needed. What we don’t see here is that the “knowledge” doesn’t have to come into consciousness. I have two cars–one with an automatic transmission, one with a stick shift. I don’t have to bring into consciousness the process for shifting gears when I get into the car that requires me to do that–I just do it!
| Types of Memory |
There are actually two basic kinds of learning and memory. One is declarative or explicit; the other is non-declarative or implicit. Knowledge of facts–what we know about places, things and people–and the meaning of these facts is explicit memory. These things must be recalled into consciousness to be used. Patients who have bilateral medial temporal lobe lesions have an inability to learn and remember items of factual knowledge. They can’t remember people that they met the day before. They can’t remember what they did the day before. Some people will further parcel explicit memories as episodic (we remember events) or semantic (we remember facts). As Kandel (2000) points out, in either case the content of all explicit memories can be expressed by declarative statements such as “I was here yesterday” (episodic) and “The hippocampus has something to do with memory” (semantic).
Implicit memory involves information about how to perform something; it’s recalled unconsciously. We use implicit memory in trained, reflexive motor or perceptual skills. I know how to drive my car; I know how to get to work. The same people with bilateral medial temporal lobe lesions can learn simple reflexive skills–they habituate and are sensitized, they can be classically and operantly conditioned (see later). They can learn certain perceptual tasks. For example, they can recall a word learned previously when given only the first few letters of the word. At the same time, they deny ever having learned the word previously. Implicit memory is often further parceled as associative and non-associative. There are two well-known types of non-associative learning: habituation and sensitization. Habituation is a decrease in response to a benign stimulus when the stimulus is presented repeatedly. A dog will be aroused when a strange tone is played. If the tone is played over and over, the dog will eventually no longer be aroused by the tone. We say that it has habituated. This kind of learning makes sense; it is not efficient for an organism to go on responding to a stimulus that has no meaning. The other form of non-associative learning, sensitization, is an enhanced response to many different stimuli after experiencing an intense or noxious one. For example, an animal responds more vigorously to a tone of lesser intensity once a painfully loud tone has been played. Here we say that the animal is sensitized.
These two forms of learning also interact. Once a response has been habituated, it can be restored by sensitization. In this case, we say that the animal is dishabituated. As an example: a habituated startle response to a noise can be restored by strongly pinching the skin.
In non-associative learning, it is not necessary that the animal learns to associate the stimuli involved (thus the name). For example, the dishabituated animal does not learn to associate the noise with the pinch. As we shall see shortly, this is the hallmark of associative learning. Not all forms of non-associative learning are as simple as habituation and sensitization. For example, we learn language by imitation of people who already speak. This involves no association of stimuli and is clearly more complicated than habituation.
In associative learning, we “learn” that two stimuli are associated with each other or that a response is associated with a given event or has a given consequence. Perhaps important in clinical considerations, a person can also learn that an outcome is not associated with a response. So a person may learn that what happens to him is not related to what he does. Two sorts of associative learning have been well studied: classical conditioning and operant conditioning. Classical conditioning is well demonstrated by Pavlov’s famous experiment in which he presented meat powder to a dog, causing it to salivate. He repeated the presentation, and each time the dog salivated. If he repeatedly rang a bell just before presenting the meat powder (they were paired), the animal came to associate the bell with the presentation of the meat powder, and it would begin to salivate when the bell was rung. In fact, for a while it would salivate if the bell was rung but no meat powder was presented (they were unpaired). After a while, the bell stopped predicting the presentation of meat powder for the dog, and it ceased salivating when it was rung. (This process is called extinction.) It should be noted that for classical conditioning to occur the ringing of the bell must precede the presentation of the meat powder, often by a certain critical interval of time (of the order of 0.5 sec). One way to look at classical conditioning is to think of the bell as becoming a signal that the meat powder is about to be presented.
In Pavlov’s paradigm, the meat powder normally elicits salivation without experimenter intervention (it is innate or perhaps previously strongly learned), and it is called the unconditioned stimulus (US). The response is called the unconditioned response (UR). The bell comes to elicit salivation only after it is repeatedly paired with meat powder; so it’s called the conditioned stimulus (CS). The response to it (again salivation) is called the conditioned response (CR). The UR and the CR are usually similar but often not identical in type or strength.
Initially investigators thought that classical conditioning involved simply learning that two stimuli were contiguous–that they occurred close together in time, one after the other. Now we think that what the animals learn is contingencies–that existence of something depends upon existence of something else. With this in mind, it is possible to see that simply learning that two stimuli were contiguous could often lead to behaviors that were maladaptive, with animals associating environmental events that had no real relationship. On the other hand, the existence of superstitious behaviors, even in humans, suggests that this does occur.
It is tempting to think of extinction as an example of forgetting, but alas it is not. The difference is that something new is learned during the process of extinction–the animal learns that the CS is no longer a signal that the US is about to occur, rather it is a signal that the US will not occur.
In operant conditioning (sometimes called trial-and-error learning), a person or animal learns that it gets a reward if it does something. So, a pigeon learns that it gets food if it pecks at a certain key, but not if it pecks at another. A rat learns that it can avoid getting an electric shock if it presses a bar at a certain time. Presumably what the animal learns is that one of its many behaviors (pecking or bar pressing) is followed by food. It is constitutional in animals to repeat behaviors that lead to positive reinforcement (something pleasant or the absence of something unpleasant) and avoid behaviors that lead to punishment or negative reinforcement.
| Neuroscience of Learning and Memory |
A great deal has been written about the kinds and properties of learning. What has been said here is probably enough for the purposes of this chapter. If you want to know more, you can consult any good textbook on learning or the psychology of learning. We want to know about what is going on in the brain when a person or animal learns something, stores what has been learned and later retrieves it for use in behavior.
Explicit Memory
In overview, experiments on learning can be interpreted to say that explicit memory is first acquired through one or more of the three polymodal association areas of the cerebral cortex, namely prefrontal, limbic and parieto-occipital-temporal. Then, the information is transferred to parahippocampal and perirhinal cortices, entorhinal cortex dentate gyrus, hippocampus, subiculum and back to entorhinal, parahippocampal and perirhinal cortex. The locations of these areas relative to one another are shown in Fig. 18-1, whereas a block diagram of the connections is shown in Fig. 18-2.
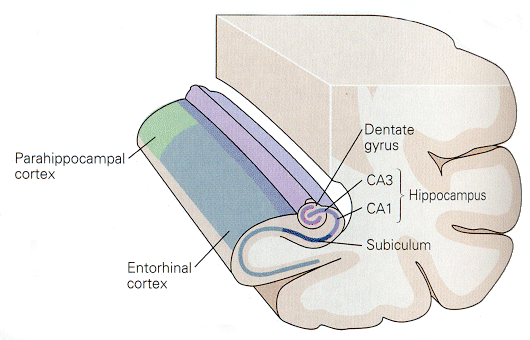 |
Fig. 18-1 - The relative positions of parts of the limbic system involved in learning and memory. (Kandel, ER, JH Schwartz and TM Jessell (2000) Principles of Neural Science. New York: McGraw-Hill.) |
Different forms of learning are affected differentially by lesions in different locations. Damage to parahippocampal, perirhinal and entorhinal cortices produces greater deficits in memory storage for object recognition than does hippocampal damage. Right hippocampal damage produces greater deficits in memory for spatial representation, whereas left hippocampal damage produces greater deficits in memory for words, objects or people. In either case, the deficits are in formation of new, long-term memory; old memories are spared.
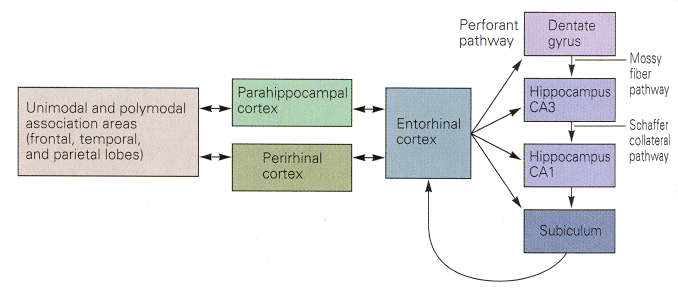 |
Fig. 18-2 - The relative positions of parts of the limbic system involved in learning and memory. (Kandel, ER, JH Schwartz and TM Jessell (2000) Principles of Neural Science. New York: McGraw-Hill.) |
Current thought is that the hippocampal system does the initial steps in long-term memory storage–different parts being more important for different kinds of memory. The results of hippocampal machinations–presumably memories–are transferred to the association cortex for storage.
There is no general semantic (factual) memory store; rather memories of a single event can be stored in multiple locations. This make sense when it is recalled that a single memory has multiple facets–each event contains sounds, smells, tastes, somatosensory experiences, visual images and so forth. Long-term storage of episodic (event) memories seems to occur in prefrontal association cortex.
So, each new explicit memory is formed by four sequential processes:
Encoding-information for each memory is assembled from the different sensory systems and translated into whatever form necessary to be remembered. This is presumably the domain of the association cortices and perhaps other areas.
Consolidation-converting the encoded information into a form that can be permanently stored. The hippocampal and surrounding areas apparently accomplish this.
Storage-the actual deposition of the memories into the final resting places–this is though to be in association cortex.
Retrieval-memories are of little use if they cannot be read out for later use. Less is known about this process.
Implicit Memory
Implicit memories are stored differently depending upon how they are acquired. “Fear conditioning” (training that involves use of fearful stimuli) involves the amygdala. Operant conditioning involves the striatum and cerebellum. For example, eye blink conditioning is disrupted by lesions of the dentate and interpositus nuclei of the cerebellum. Classical conditioning, sensitization and habituation involve the sensory and motor systems involved in producing the motor responses being conditioned. Perhaps surprisingly, certain simple reflexes mediated by the spinal cord can be classically conditioned even after the cord has been surgically isolated from the brain. So, it appears that all regions of the nervous system may be capable of memory storage.
| Processes of Learning |
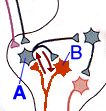 |
Fig. 18-3 - A reverberating circuit: Neuron A excites B and vice versa. (Kandel, ER, JH Schwartz and TM Jessell (2000) Principles of Neural Science. New York: McGraw-Hill.) |
Given the definitions for learning and memory, what sort of mechanisms would we expect to find in the nervous system? One early thought was that neurons in “memory” pathways were arranged in reverberating circuits. In such a circuit, one neuron excites another and the other excites the one such that, once the circuit is activated, action potentials run around continuously. An example of this kind of arrangement is shown in Fig. 18-3. Here are shown only 2 neurons in the circuit but any number may be included. If this kind of arrangement accounts for memory, then any event that temporarily stopped activity in the circuit should disrupt memory. Unfortunately for supporters of the idea, electroconvulsive shock, which temporarily stops or resets all electrical activity in the nervous system produces only a significant, transitory loss of recent memory, but no loss of older memories.
Some years ago, the psychologist Donald Hebb (Hebb, DO (1949) The Organization of Behavior: A Neuropsychological Theory. New York: John Wiley) mulled this problem and came up with a principle that has become known as Hebb’s rule. Briefly, the principle is “When an axon of cell A . . . excites cell B and repeatedly or persistently takes part in firing it, some growth process or metabolic change takes place in one or both cells so that A’s efficiency as one of the cells firing B is increased.” As we shall see, current thought is an extension of Hebb’s rule.
Habituation
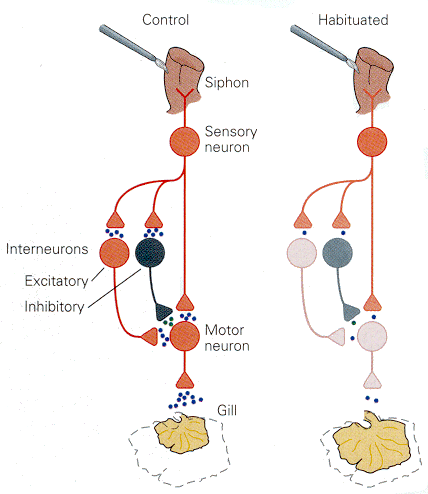 |
Fig. 18-4 - Simplified neural circuits involved in the habituation process in Aplysia. There are about 24 sensory neurons in the siphon; these are glutaminergic. They synapse on 6 motor neurons that innervate the gill and various interneurons as shown. The control condition is shown on the left, the habituated condition on the right. (Kandel, ER, JH Schwartz and TM Jessell (2000) Principles of Neural Science. New York: McGraw-Hill.) |
What happens in the nervous system to produce habituation? Experiments performed in Aplysia californica, the sea slug, were designed to address this problem. Their results are shown schematically in Fig. 18-4. If the siphon of the animal is stimulated mechanically the animal withdraws the gill, presumably for protection. That action is known to occur because the stimulus activates receptors in the siphon, which activates, directly or indirectly through an interneuron, the motoneuron that withdraws the gill. This is a simple reflex circuit. All of this is shown on the left side of the figure. With repeated activation, the stimulus leads to a decrease in the number of dopamine-containing vesicles that release their contents onto the motoneuron. There appears to be no change in the sensitivity of postsynaptic NMDA or non-NMDA receptors. As yet, we don’t know why the dopamine release decreases. It is presumed that habituation in vertebrates, including man, occurs by a similar process.
Sensitization
In sensitization, a stimulus to one pathway enhances reflex strength in another. An example, again taken from experiments in Aplysia, is shown in Fig. 18-5. Again, stimulation of the siphon leads the animal to withdraw the gill by activating sensory neuron 1, which in turn activates a motoneuron. If the tail of the animal is stimulated just before the siphon is, then the withdrawal of the gill is quicker and more forceful. The mechanism of this appears to involve serotoninergic, axo-axonic synapses. As shown in the figure, activation of the sensory receptors in the tail activates, through sensory neuron 2, a facilitating interneuron that excites sensory neuron 1 in the pathway leading the gill withdrawal. It does this either at the cell body or at the terminals of the sensory neuron on the motoneuron or the interneuron. The consequence of the sensitization process is to increase the size of the EPSP in the motoneuron without increasing the response of sensory neuron 1. This will cause a greater response in the motoneuron and therefore a greater withdrawal of the gill.
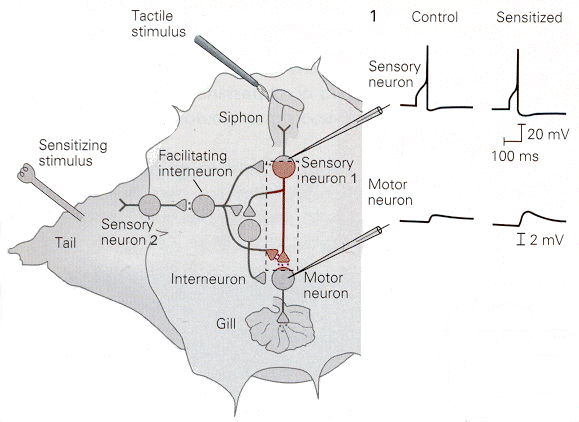 |
Fig. 18-5 - Sensitization is produced by applying a noxious stimulus to the tail of the Aplysia's tail, activated sensory neuron 2. This, in turn activates a facilitating interneuron that enhances transmission in the pathway from the siphon to the motor neuron. (Kandel, ER, JH Schwartz and TM Jessell (2000) Principles of Neural Science. New York: McGraw-Hill.) |
How all this occurs is illustrated in Fig. 18-6, which shows an axo-axonic synapse as might occur between the facilitating interneuron and sensory neuron 1. Serotonin (5-hydroxytryptamine or 5HT) is released by the presynaptic axon onto the postsynaptic axon where it binds to receptors and activates a G protein that, in turn, activates adenylyl cyclase to produce cAMP. This cAMP activates a cAMP-dependent protein kinase, PKA. Along with another kinase, PKC, PKA phosphorylates and closes K channels (hypopolarizing the cell), mobilizes vesicles for exocytosis and opens Ca channels. The end result is that activation of this 5HT pathway by tail stimulation causes more transmitter substance to be released by siphon stimulation, the resulting larger EPSP leads to a larger response by the gill.
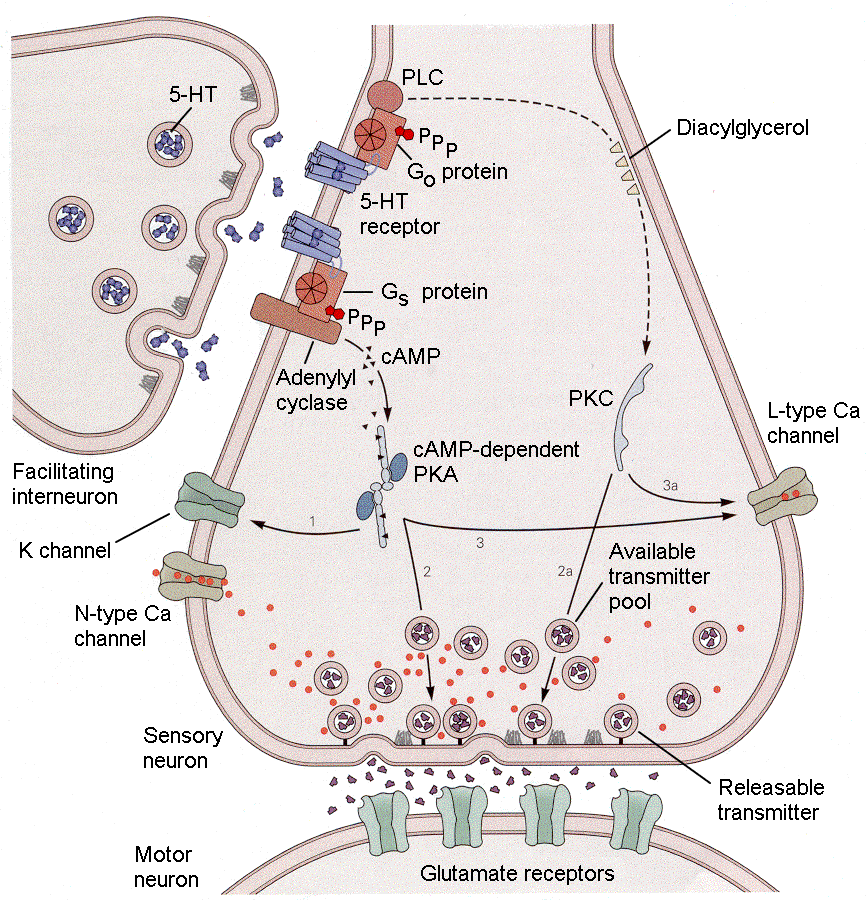 |
Fig. 18-6 - The synaptic and chemical events underlying presynaptic facilitation involved in producing sensitization. See text for details. (Kandel, ER, JH Schwartz and TM Jessell (2000) Principles of Neural Science. New York: McGraw-Hill.) |
With only short-term tail stimulation, the sensitization will fairly quickly disappear when tail stimulation ceases. However, the sensitization can be made relatively permanent by repeated tail stimulation. This long-term sensitization (and also long-term habituation) occurs because there are structural changes that occur in the presynaptic terminals (sensory neuron 1, for example). With sensitization, there is an up to 2-fold increase in the number of synaptic terminals in both sensory and motoneurons. Alternatively, with habituation, there is a one-third reduction in the number of synaptic terminals. Both of these changes require altered protein synthesis by mechanisms shown in Fig. 18-7.
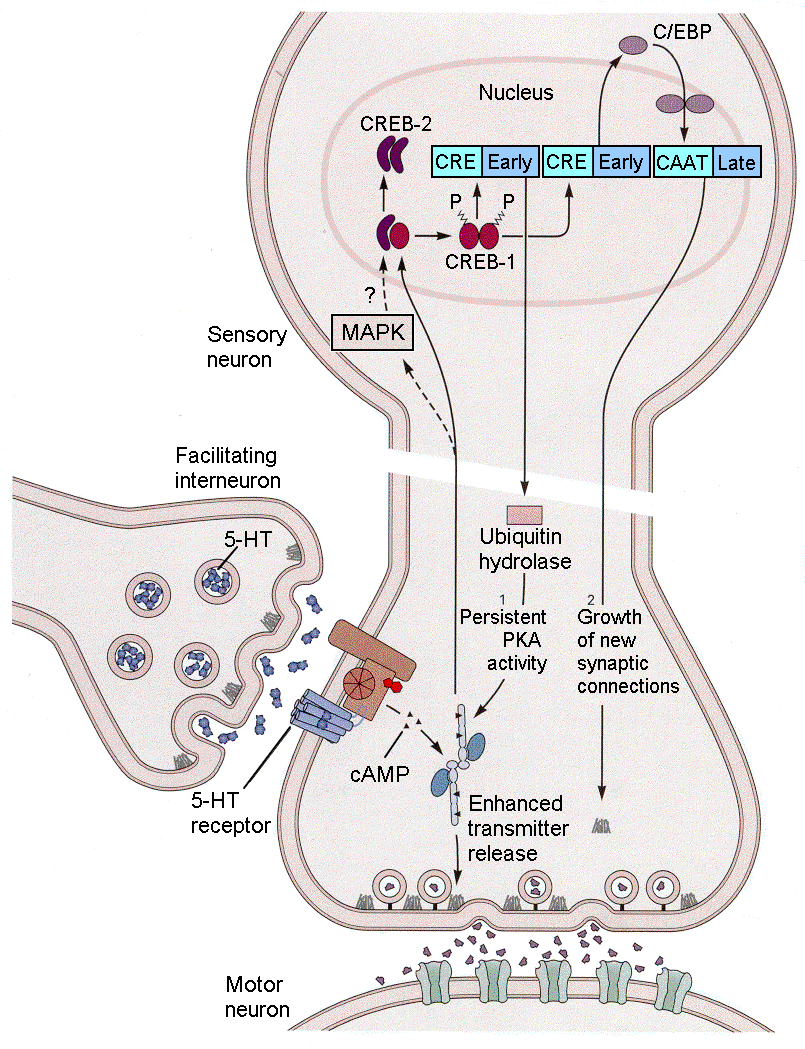 |
Fig. 18-7 - Long-term storage of implicit memory for sensitization involves changes shown in Fig. 18-6 plus changes in protein synthesis that result in formation of new synaptic connections. (Kandel, ER, JH Schwartz and TM Jessell (2000) Principles of Neural Science. New York: McGraw-Hill.) |
Long-term Potentiation
As previously detailed, the hippocampus is important in storage of declarative memory. In 1973, a phenomenon was described in the hippocampus that may account for declarative memory. Since then the same phenomenon has been observed in various other places known to be involved in memory storage. This phenomenon is called long-term potentiation (LTP).
A high-frequency train of stimuli applied to fibers afferent to the hippocampus increase the amplitude of EPSPs in the target neurons. The increase lasts for days or weeks and requires activation of several afferent axons together. This property has been termed cooperativity, and it results from the requirement of NMDA receptors that glutamate bind to them and the cell be hypopolarized, the binding opens the channel and the hypopolarization displaces Mg++ that blocks the channel lumen. Also required is that the pre- and postsynaptic cells both be active at the same time. This latter property has been termed associativity. The astute student will see that this is precisely the condition that Hebb’s law says should exist.
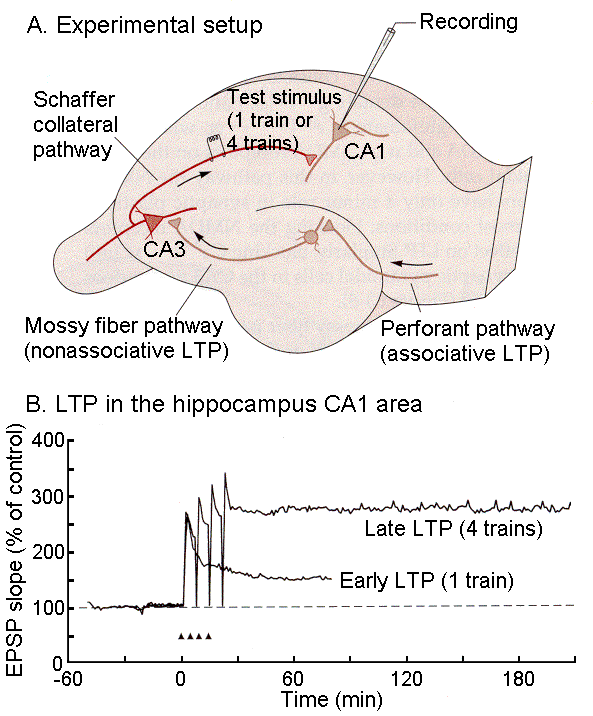 |
Fig. 18-8 - A. Experimental setup for demon-strating LTP in the hippocampus. The Schaffer collateral pathway is stimulated to cause a response in pyramidal cells of CA1. B. Comparison of EPSP size in early and late LTP with the early phase evoked by a single train and the late phase by 4 trains of pulses. (Kandel, ER, JH Schwartz and TM Jessell (2000) Principles of Neural Science. New York: McGraw-Hill.) |
The experimental setup for demonstrating LTP is shown in Fig. 18-8A. Recordings are made intracellularly from CA1 neurons of the hippocampus while stimulation is applied to the Schaffer collaterals of CA3 neurons. The amplitudes of the EPSPs in the CA1 neurons are shown in B. For a single stimulus, the amplitude of the EPSPs is plotted at 100%. When a train of stimuli is applied instead, the amplitude of the EPSPs augment to about 150%, whereas with 4 such trains the amplitude increases to 250%. Many people think that long-term potentiation is an example of Hebb’s rule at work and that it is the physiological basis of memory.
During normal synaptic transmission (Fig. 18-9), glutamate binds to non-NMDA receptors allowing cations to flow through the channels and the cell membrane to hypopolarize. Glutamate also binds to metabotropic receptors, activating PLC, and to NMDA receptors. As you may already know, NMDA receptor channels can bind glutamate but no current will flow through the channels unless the Mg++ that binds to the channel lumen is displaced. The latter event can be effected by hypopolarizing the cell.
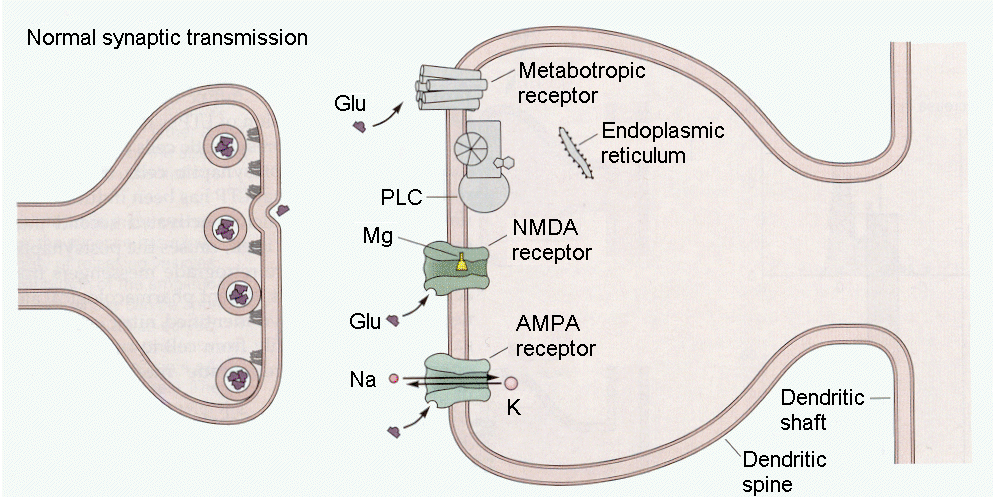 |
Fig. 18-9 - During normal low-frequency trans-mission, glutamate interacts with NMDA and non-NMDA (AMPA) and metabotropic receptors. (Kandel, ER, JH Schwartz and TM Jessell (2000) Principles of Neural Science. New York: McGraw-Hill.) |
By contrast, during the early phase of LTP, the high-frequency stimulation opens non-NMDA glutamate channels leading to hypopolarization. This dislodges Mg++ from the NMDA glutamate channels, and Ca++ enters the cells. The calcium triggers the activity of Ca-dependent kinases, PKC and Ca-calmodulin, and tyrosine kinase. Ca-calmodulin kinase phosphorylates non-NMDA channels, increasing their sensitivity to glutamate and a messenger is sent retrogradely to the presynaptic terminal to increase the release of transmitter substance. All of this is illustrated in Fig. 18-10. These events increase the transmitter released by presynaptic terminals. With LTP, there is a decrease in transmission failure, i.e., synapses are more reliable in exciting postsynaptic cells. This is also shown in the figure.
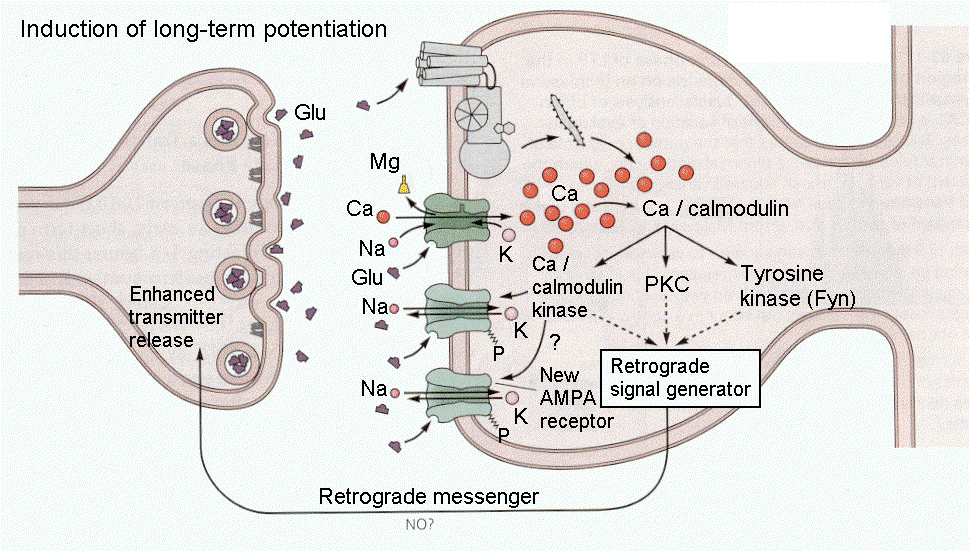 |
Fig. 18-10 - With high-frequency stimulation other events occur as described in the text. (Kandel, ER, JH Schwartz and TM Jessell (2000) Principles of Neural Science. New York: McGraw-Hill.) |
In the late phase of LTP (Fig. 18-11), calcium enters the cell and triggers Ca-calmodulin, which in turn activates adenylyl cyclase and cAMP kinase. The latter translates to the nucleus of the cell and starts processes that lead to protein synthesis and to structural changes, i.e., the formation of new synapses. Many scientists believe that this is the substrate for long-term memory–the formation of new synapses.
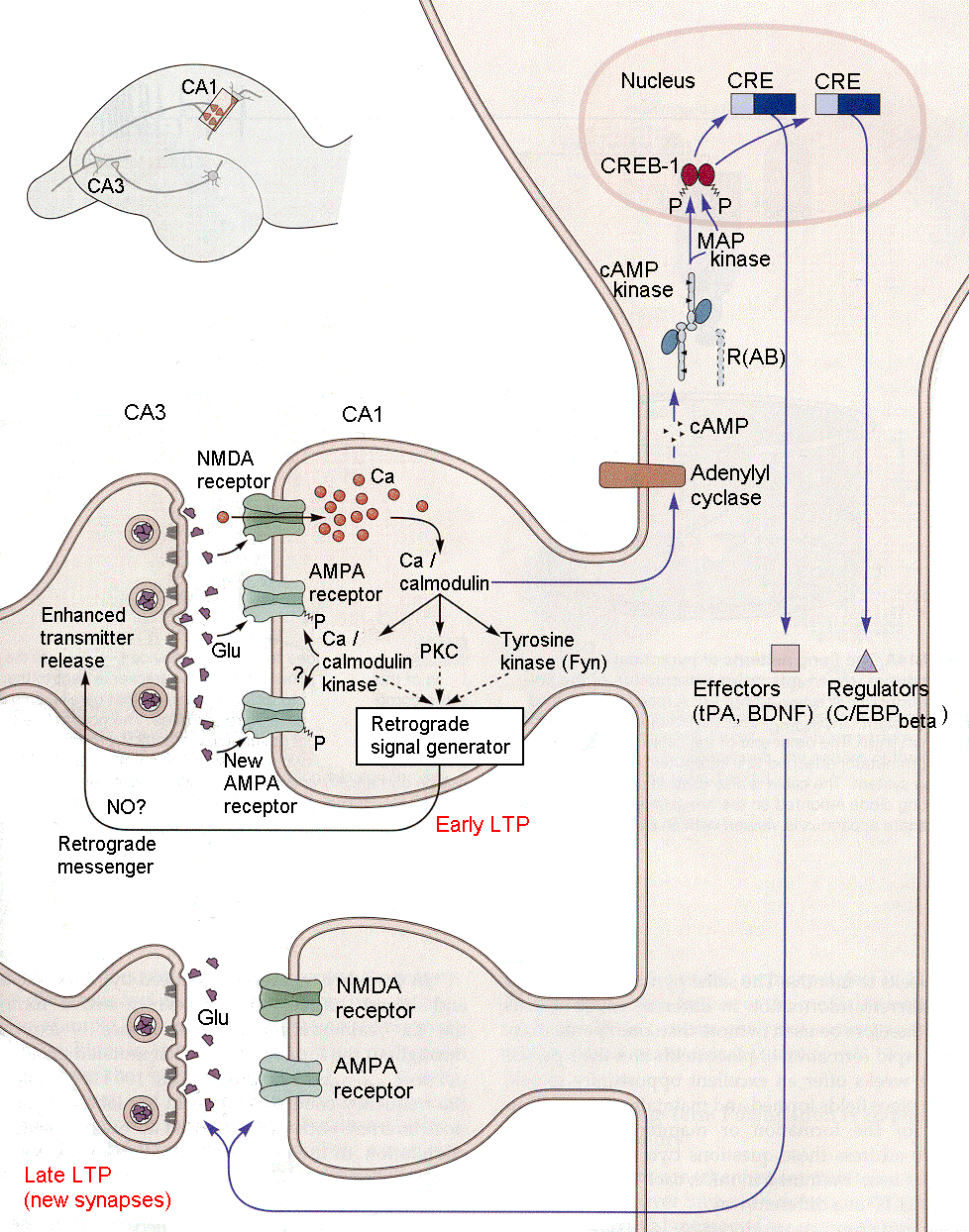 |
Fig. 18-11 - For LTP to last (Late LTP) the events of Fig. 18-10 must also lead to changes in protein synthesis and to formation of new synaptic connections. (Kandel, ER, JH Schwartz and TM Jessell (2000) Principles of Neural Science. New York: McGraw-Hill.) |
There are still unanswered questions about the relationship of LTP to memory. First, memories last decades whereas LTP has been observed only for days or weeks. How long LTP can be maintained is difficult to determine. Admittedly, LTP is the longest lasting process known in neuroscience. Still memories may last much longer. LTP occurs in most or all of the places where memories are known to be stored. What is not known is whether disruption of LTP also interferes with memory.
| Summary |
Non-declarative (implicit) memory involves different brain regions: fear conditioning involves amygdala; operant conditioning involves striatum and cerebellum; and classical conditioning, sensitization and habituation involve sensory and motor systems used in the responses. This kind of memory involves a number of processes: habituation involves decrease in synaptic strength from decreased transmitter release; sensitization involves increase in synaptic strength due to presynaptic facilitation; and classical conditioning involves increase in synaptic strength due to presynaptic facilitation that is dependent on activity in both pre- and postsynaptic cells.
Declarative (explicit) memory also involves a number of brain regions: there is no general store for explicit memories; because the subject of memories is multimodal, storage of different aspects occurs in different locations; the hippocampal formation is important in processing information for storage as memory; and memories are actually stored in association cortex. This kind of memory probably makes use of long-term potentiation. The early phase of LTP involves glutamatergic transmission; postsynaptic processes that produce enhanced sensitivity or receptors to glutamate as well as enhanced release of transmitter substance. In the late phase of LTP, protein synthesis leads to changes in cell structure and formation of new synapses.
| Suggested Reading |
- Dudai, Y (1989) The Neurobiology of Memory: Concepts, Findings, Trends. Oxford: Oxford University Press.
- Hebb, DO (1949) The Organization of Behavior: A Neuropsychological Theory. New York: John Wiley
- Hull, CL (1943) Principles of Behavior. New York: Appleton-Century-Crofts.
- Kandel, ER, JH Schwartz and TM Jessell (2000) Principles of Neural Science. New York: McGraw-Hill.
- Kandel, ER and JH Schwartz (1982) Molecular biology of learning: Modulation of transmitter release. Science 218:433-443
- Kimble, GA (1961) Hilgard and Marquis’ Conditioning and Learning. 2nd Edition. New York: Appleton-Century-Crofts.
- Nicoll, RA, JA Kauer and RC Malenka (1988) The current excitement in long-term potentiation. Neuron 1:97-103.
- Sargent, SS and KR Stafford (1965) Basic Teachings of the Great Psychologists. Garden City, NY: Dolphin Books.