|
Emotional behavior
Emotion is another of the behaviors for which there is really no satisfactory definition. It has
both unobservable internal and observable external aspects. It is a cognitive process in that one
must perceive the causative situation and evaluate it in light of past experience and cultural
variables. For example, a particular situation can be potentially beneficial or deleterious to one's
self, family, or country. The person may not even be aware that the evaluation is occurring.
Emotion is either pleasant or unpleasant and need not be the same to every individual. It is
expressed outwardly in the form of facial expressions, gestures, vocalizations, body postures, and
other movements, or it may be expressed in the form of an absence of movement. There may be
concomitant autonomic activity resulting in changes in heart rate, blood pressure, piloerection,
sweating, and flushing. Movements of
emotion can propel the individual toward
another object, as in attacks, or away, as
in flight. Finally, emotional behavior can
result in excitement and alertness or
depression, dullness, and sluggishness.
The initiation and regulation of
emotional behavior by the brain is
thought to involve the limbic system.
Knowledge of the limbic system is quite
old; it was named by the surgeon, Broca,
in the last century. Actually, little
research was done to link the limbic
system with emotion until about 1937
when the neuroanatomist, Papez, published a proposed mechanism of emotion involving the
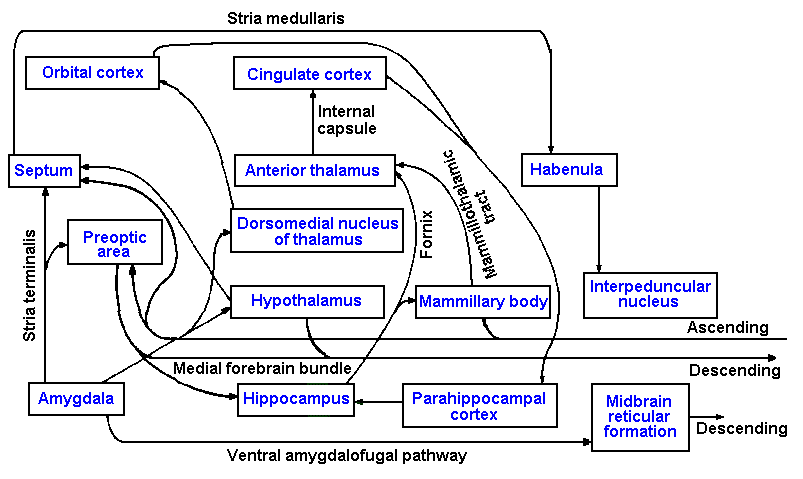
limbic system. Figure 17-1 is a summary diagram of most of the parts of the limbic system and
their interconnections. The word limbic means "in a form of a ring." The system is given this
name because the various parts are organized into rings of interconnected structures. The major
parts of the limbic system are as follows: (1) structures of the temporal lobe of the cortex: the
amygdala, hippocampus, and parahippocampal cortex; (2) other cortical structures: the orbital and
cingulate cortices; (3) anterior and dorsomedial thalamic nuclei; (4) the hypothalamic structures:
the hypothalamus proper, septum, and preoptic area; (5) the mammillary bodies; (6) the habenula;
(7) the interpeduncular nuclei; and perhaps others. The limbic system receives input from
enteroceptors and exteroceptors. It is intimately related to the olfactory system, and it also
receives inputs from every sensory modality. It sends its output to various autonomic nuclei in
the brain stem and, through the hypothalamus, influences every part of the body through release
of hormones from the pituitary gland.
It has already been noted that removal of structures of the hemisphere above the mammillary
bodies results in sham rage, a rage that is typical of normal aggressive behavior except in its lack
of direction toward the offending object. The rage is most intense if the lesion is made
immediately above the mammillary bodies, less if the hypothalamus is spared, and least if only the
neocortex is removed. If a lesion is made in the ventromedial hypothalamic nuclei, a similar rage
results but, unlike sham rage, the attacks are well directed. Similarly, a lesion in the septal area
makes a normally placid animal savage. Interestingly, a subsequent lesion of the amygdala makes
the animal tame again. Bilateral removal of the amygdala without a concomitant septal lesion
leads to decreased aggressive behavior in normally savage animals.
Stimulation in the ventromedial and other areas of the hypothalamus elicits aggressive or
attack behavior or what appears to be stalking and prey-killing behavior in animals that begins
when the stimulus is turned on and ends when it is turned off. Aggressive behavior is also elicited
by stimulation in the amygdala, but, unlike that due to hypothalamic stimulation, it develops more
slowly and outlasts the stimulus by some period of time. In addition to aggressive behavior,
amygdalar stimulation also leads to cessation of movements, changes in spinal reflex activity,
controversive head and eye movements, swallowing, licking, chewing, changes in heart and
respiration rates, changes in gastrointestinal motility, micturition, defecation, pupillary dilation,
and piloerection. These changes are all characteristic of emotional responses, though not
necessarily of aggressive behavior. In humans, stimulation of the amygdala leads to sensations of
fear, disturbances of mood, a sense of unreality, and distortions of bodily perceptions.
Bilateral removal of the temporal lobe leads to a constellation of effects that are called the
Kluver-Bucy syndrome. The symptoms include the following:
- Visual agnosia-inability to recognize seen objects
- Compulsive exploratory behavior-everything is examined with the mouth or by smell
- Passivity, unresponsiveness, and decreased emotional responsiveness
- Lack of fear
- Intensification of sexual activity-increased frequency and directed toward the same sex or
even different species
- Changes in dietary patterns
Attempts have been made to localize the structures whose removal produced each of these
symptoms. There has been little success in such efforts.
Stimulation in the region of the medial forebrain bundle or the preoptic area produces a
sensation described as pleasurable by humans. Apparently, animals also find it pleasurable
because they will press a lever in order to receive an electrical stimulus in this area, some people
call the pleasure center. They will elect to press the lever and receive the shook rather than eat
when they are hungry, drink when they are thirsty, or engage in sexual activity with a receptive
female. In fact, rats will cross an electrified grid that they normally would not cross, in order to
press the lever and receive the shock. Obviously, the sensation is a powerful one in influencing
behavior.
It is probable that the hypothalamus cannot, by itself, initiate directed emotional behavior, but
requires the modulating, directing, and regulating influence of the rest of the limbic system. As
we have seen, stimulation of various parts of the limbic system can activate all of the autonomic
and behavioral concomitants of emotional behavior, but as yet we do not know how or where the
behavior is initiated or what parts of the system play what roles in what behaviors. It is clear that
the limbic system plays an important role in emotional behavior.
Learning and memory
Learning is usually defined as a relatively permanent change in behavior as a result of practice
or experience. There is a vast psychological literature dealing with the kinds of learning and their
characteristics, which we will not attempt to go into here. Animals can learn certain responses to
stimuli just by associating the stimuli with other stimuli that normally elicit the desired response
(classical conditioning). For example, if an animal is presented with a puff of air directed at the
eye, it blinks. Repeated pairings of a brief light flash or tone and a puff of air effect some change
in the nervous system such that the light flash or tone, by itself, evokes the blink. Another kind of
learning is called instrumental or operant conditioning. In instrumental conditioning, an animal
must perform some task in order to get a reward or avoid punishment. Thus, an animal will learn
new behaviors in order to get a reward or relief from pain. This phenomenon is familiar to every
animal trainer. We now know that animals will learn complex behavior patterns in order to
receive electrical stimulation in parts of the hypothalamus, particularly the preoptic area, the
pleasure center.
Some theorists claim that human learning can be explained in terms of classical and
instrumental conditioning, whereas others maintain that there is something more, something
unique about human learning ability. This something, if it exists, has yet to be identified. Much of
what we know about learning has been obtained from animals, raising the question of the
applicability of the results to humans. It has already been noted that humans may differ even from
monkeys in the indispensability of the cerebral cortex in sensory functions, so it should be
cautioned that there may be profound differences between man and animals, even in elementary
learning processes. Because animal evidence is all that is available in many cases, we will treat it
as if it also applied to humans.
In general, the following principles seem to emerge from studies on learning:
- Ablation of the cerebral cortex has no effect on classical conditioning. The results of
early studies were taken to indicate that classical conditioning could not occur in the
absence of cerebral cortex; we now know this is untrue. Classical conditioning can be
demonstrated in a part of the spinal cord isolated by transection, indicating that even the
spinal cord is capable of this sort of learning.
- If the task to be learned requires a sensory capacity that is abolished by a lesion in the
cerebral cortex, then no amount of training is successful in teaching the task. For
example, some visual discrimination learning requires a visual capacity that is abolished
by a lesion of the visual cortex. Visual discrimination learning of this kind is impossible
following such a lesion. (However, recent evidence suggests this point may have to be
reexamined. Look back at Chapter 7.)
- Retention is impaired, but retraining is possible for instrumental conditioning if lesions
(that do not abolish a needed sensory capacity) are made in primary cortical sensory
areas. For example, the primary somesthetic area is not required for somesthesia, and
somatic discrimination learning is possible following a lesion to this area. On the other
hand, it is known that parietal lobe lesions (not necessarily restricted to the primary
somesthetic area) interfere with the ability of humans to discriminate the shape of
objects either by manipulating them (astereognosis) or by viewing them. Attempts to retrain this ability have failed.
- If a number of sensory modalities is involved in the learning process, then a bigger
cortical lesion is needed to interfere with learning than if a single modality is involved.
In fact, the amount of impairment is a function of the amount of cortical tissue removed
and is independent (to some extent) of where on the hemisphere it is removed. This has
been termed the law of mass action. This would follow whether learning were a
localized function or were distributed across the surface of the cortex.
- Lesions that involve both primary sensory cortex and association cortex of one modality
may cause considerable, lasting impairment of learning in that modality (a notable
exception is the modality of smell).
- The temporal pole and the junction between temporal and occipital cortices seem to be
important for learning visual discrimination tasks.
- Prefrontal cortex seems to be involved in learning ordered responses where time is a
critical feature. This is seen most readily in delayed response learning, where the person
or animal is required to delay a response until some time after the cues are presented.
Monkeys with prefrontal lesions can find a raisin they have seen placed in a covered
well if they are allowed to do so within a short time, but they cannot, if they are
required to wait more than a few minutes. This is an easy task for normal monkeys.
- When learning involves the cortex on one side only (e.g., when cues are presented in
only half of the visual field), the corpus callosum participates in transfer to the other
cortex, at least for some learned responses. If the optic chiasm is transected
midsagittally, the image from each eye is transmitted only to the ipsilateral hemisphere.
After such a lesion, an animal can perform a visual discrimination task learned with one
eye covered, using either eye to sense the objects. If a transection of the corpus
callosum is done with the transection of the optic chiasm, then the task can be
performed only with the eye uncovered during training. The other eye can be used to
relearn the task. Apparently, the learning normally occurs bilaterally by way of the
corpus callosum.
With instrumental conditioning, animals can change the behavior of neurons in the same way
they change their overt behavior. These changes in the discharges of neurons, as a result of
conditioning, could be brought about by making or changing some overt movement or behavior.
We know that neurons in the CNS are responsive to sensory input from the periphery, sometimes
evocable by an appropriate movement. A neuron, responsive to stimulation of a particular area of
skin, may discharge if the animal brings the area into contact with some object, even its own body.
In fact, in some of these conditioning experiments, movements or changes in movements have
been observed concomitant with changes in neural activity. Clearly, the animal could be
stimulating itself. It is also conceivable that a neuron's discharge could be altered by itself, that is,
in isolation from any movement or sensation. Evidence suggests that this actually does not
happen (Wyler, Burchiel, Robbins, 1979), but, because there is nearly always some muscle
contracting somewhere in the body, this is a difficult demonstration. Work is in progress to find
some way to use this control over nerve cells to run prosthetic devices for lost limbs. As yet, the
work has just begun.
Attempts have been made to record both evoked potentials and discharges of single cells or
small groups of cells during training of animals in order to find neural concomitants of learning.
There are changes in evoked potentials that occur while an animal is learning a particular task.
Unfortunately, these changes are difficult to interpret for several reasons. It is not known what
events (postsynaptic potentials) are responsible for producing the various parts (positive and
negative deflections) of the evoked potential, if, indeed, a single event is responsible for each part.
Because the evoked potential reflects activity in all neurons neighboring the recording electrodes,
we cannot say with certainty which neurons we are studying. Learning has been defined as a
change in behavior; usually this means a change in movement. We cannot, as yet, separate the
neural events, parts of an evoked potential, that are associated simply with the production of a
movement from those associated with the learning process itself.
Interpretations of single-cell recordings during learning are even more difficult. The problem
of separating changes due to motor activity from changes due to learning itself is also inherent in
single-cell recordings. In addition, we cannot follow the behavior of a single cell long enough to
observe it both before and after learning has occurred, and, over such a long period, we could not
be sure it was, in fact, the same neuron. The neurons that are near the recording electrode after
learning has occurred may behave differently either because they have "learned" or because they
are different neurons or because they are damaged by the recording electrode.
Memory is the ability to recall events or tasks learned or experienced minutes, days, or years
before. It is customary to divide memory into two types, immediate recall or short-term memory and long-term memory. Short-term memory usually has a duration of minutes to
perhaps hours, whereas long-term memory has a duration from minutes (or hours) to years. It is
not possible to say precisely when a given memory crosses the border from short-term into long-term memory or even if short- and long-term memory involve the same continuous process or two
different processes.
Animals given electroconvulsive shocks or other treatments to interrupt brain activity within
about 5 minutes after a training session do not remember the task they were trained to perform
when tested some time later. When no treatment is given, the same animals remember the task 5
minutes and even weeks after the training sessions. If the same treatment is given to the animal 4
hours after training sessions, it has no effect at all on their ability to remember the task even
weeks later. It thus appears that there is a period shortly after an event when the memory is
vulnerable to disruption; then something happens to make the memory trace resistant to erasure.
It is this event that constitutes the conversion from short-term to long-term memory.
Clinically, it is found that immediately following electroshock therapy or some brain
concussions there is no memory of the therapy or the concussion or of events that occurred some
time preceding it. In man, this lack of memory or amnesia may encompass longer periods than in
animals, sometimes day, weeks, or even years, but more remote memories are usually intact. The
patient, under these circumstances, is still capable of forming new long-term memories, but there
are conditions where this ability is also impaired.
The amnesic syndrome, or Korsakoff's psychosis, is an impairment of the ability to convert
short-term memory into long-term memory, without impairment of immediate recall and without
elimination of already formed long-term memories, and it results from bilateral damage to the
hippocampal formation of the temporal lobes. Some pathology is usually also found in the
mammillary bodies, a major projection site of the hippocampus. The uncus, parahippocampal
cortex, mammillary bodies, and amygdala do not seem to play a role in this syndrome, nor do
bilateral lesions of the fornix, the output pathway from the hippocampus, produce impairment of
memory. The patient can recall bits of information for minutes (immediate recall), but if he is
distracted these are lost. He can also recall events that happened prior to the damage to his brain
(long-term memory).
The amnesic syndrome can sometimes result from thiamine deficiency or bilateral occlusion of
the posterior cerebral arteries that supply the hippocampus. It also results from surgical removal
of one temporal lobe (temporal lobectomy) when the other has been damaged. Temporal
lobectomy without prior damage to the other hippocampus does not produce any memory
impairment. General symptoms of the amnesic syndrome are indicated in the following case
history:
| A 29-year-old patient had been incapacitated by intractable seizures since the age of 16. As a
treatment, the mesial surfaces of both temporal lobes were resected to about 8 cm posterior of temporal pole.
Following this surgery, the patient no longer experienced seizures, but he also no longer recognized the
hospital staff nor could he find his way around the hospital. He seemed not to be able to recall the day-to-day
events of his hospital stay. He did not recall the death of his favorite uncle three years earlier, but could recall
some trivial events just before his admission to the hospital. His earliest memories were clear and vivid. Even
after three years, this patient was unable to remember a new address (though he remembered the old one) or
where he had put objects he habitually used. There was no decrease in his IQ, in fact, there was a slight
increase. His understanding and reasoning were normal, as were psychological test results for perception,
abstract thinking, motivation, and personality. He could retain a three-digit number or a pair of unrelated
words for several minutes; if he was distracted they were immediately forgotten. (Scoville WB, Milner B: J
Neurol Neurosurg Psychiat 20:11-21, 1957). |
The memory storage process apparently depends upon the integrity of the hippocampus, but
we know little about the location of long-term storage of information. Some cases of temporal
lobe epilepsy exhibit seizures preceded by auras consisting of memories of past events. Typically,
the same past event is played back to the patient just prior to each seizure, serving as a signal that
the seizure is impending. During surgery to excise the focus (the pathological cortical area
responsible for the seizure), the cortical surface of the temporal lobe is stimulated electrically to
locate the site of the focus. When the site is located and stimulated, the memory of the aura is
played back again to the patient in an interesting way. When the stimulating current is turned on,
the memory begins to be recalled by the patient, and the events in the memory continue in
sequence, as they originally happened, until the current is switched off. If, after a moment, the
stimulus is turned on again, the memory is picked up again where it left off before. It is as if a
tape recorder were being turned on and off. Removal of the focus abolishes the seizures with
their auras, but does not eliminate the memory that was part of the aura. It is unknown whether
the memories remain because of bilateral storage at conjugate sites (the site of the focus and the
same site on the contralateral hemisphere) or whether the memories are actually stored elsewhere.
No single site for storage of memory traces has yet been found in the central nervous system.
The nature of the storage process is as much a mystery as its location. It is clear that long-term memory is not a result of activity in reverberating circuits (circuits in which activity is
transmitted continuously around a closed-loop pathway), but short-term memory may be.
Electroconvulsive shock, deep coma due to overdoses of sedative drugs, and complete cessation
of electrical activity, all of which would stop reverberation, do not interfere with already formed
long-term memories, but they do destroy short-term memories and disrupt their conversion to
long-term memories. Many claims have been made about the involvement of specific, single
ribonucleic acids (RNA) or other specific proteins in memory, but in general the claims remain
unsubstantiated. It is noteworthy that drugs that block protein synthesis will also block
transformation of short-term into long-term memory. Also, a variety of CNS stimulants have
been shown to improve learning in animals and restore learned behaviors that have been lost as a
result of CNS lesions. The process must certainly involve biochemical changes in cells, perhaps in
formation of new or more permanent or more effective synaptic junctions.
Personality
It has been known for over 100 years that the prefrontal lobes are involved in determining and
regulating personality. Seizures brought on by abnormalities of the prefrontal lobes often involve
transient changes in personality, with compulsive behavior and the release of social inhibitions that
may persist for several weeks following the seizures. Bilateral removal of the prefrontal lobes
results in a reduction in emotional responsiveness and anxiety, but no change in perception. For
example, patients with prefrontal lobotomies still report feeling pain, but they say it no longer
bothers them. The patients become impulsive and distractible; they lack concern over the
consequences of their actions; and they are unable to plan ahead. Following lobotomy, a man
urinated in the corner of the room. When told not to repeat this action, he agreed, but shortly
was back in the corner again. A woman, who had planned and prepared meals for her family for
many years, was unable to do so following a lobotomy, although she was able to perform the
individual activities involved when told what to do next.
Prefrontal lobotomy patients sometimes have difficulty postponing gratification. Although
they are distractible, they show a tendency to persevere in activities and seem unable to shift their
responses to suit a change in environment or circumstance. Their thinking often becomes rigid
and concrete with some deficits in abstract reasoning. These symptoms are illustrated in the
following case history:
| A 60-year-old housewife and fashion designer was admitted to the hospital with a history of several
years of fatigue, loss of ambition, lack of enthusiasm, and a tendency to readily cry over minor problems. A
depression of mood and a tendency toward introspection had also developed. She also reported loss of appetite
and weight. Neurological examination was normal except for slowness of speech and thought processes.
Thyroid function tests indicated hypothyroidism, for which she was treated and released.
After discharge, the emotional and personality symptoms continued and worsened. She also developed
difficulty in walking, unsteadiness of gait, difficulty climbing stairs, and getting out of chairs. Her motor and
mental activities became slower and she procrastinated in making decisions. There was a progressive deficit
in memory and episodic blurring or fogging of vision.
At age 67, she was readmitted to the hospital with slowness of speech and gait, slight disorientation for
date and year, exaggerated deep tendon reflexes, and no significant sensory findings, except a slight decrease
in position sense of the toes. Shortly, she developed headaches, vomiting, increasing somnolence, a Babinski
sign, and sluggish pupillary responses. Cerebrospinal fluid pressure was elevated, as was fluid protein.
Bilateral carotid arteriogram revealed a butterfly-shaped meningioma in the anterior cranial fossa that was
removed. Histological examination indicated that the tumor was benign. The patient showed immediate
improvement at the conclusion of surgery. Pupils were equal in size and reactive to light. Two months later
she could recognize the doctor by name, but did not know the place or time. Assistance was still required
when walking. There was also some impairment of recent memory. The development of symptoms indicated
an involvement of prefrontal lobes by the tumor, compressing them and producing alterations of mood and
personality. Continued growth of the tumor compromised premotor and perhaps motor areas resulting in
disturbance of gait, possibly due to compression of the anterior cerebral arteries, and later possibly also the
temporal lobes were compromised resulting in memory loss. (Curtis BA, Jacobson S, Marcus EM:
Introduction to the Neurosciences. Philadelphia, Saunders, 1972) |
In the 1940s and 1950s, prefrontal lobotomies were frequently performed in mental
institutions and prisons on patients whose behavior was considered undesirable and intractable.
The procedure made them docile and carefree, but the alterations in their personalities were
severe. At one point, it became so fashionable to have a lobotomy performed that it actually
became an office procedure. Only a small incision in the orbit above the eye is required. Some
physicians did as many as 1000 leukotomies per month during one period. Fortunately, anxiety
and intractable behaviors are now controllable on a temporary basis by the use of tranquilizers,
and lobotomies are seldom performed. Some states allow them only in cases of uncontrollable,
incapacitating anxiety or fear or for relief of the suffering from pain in terminal illness, where it
has been shown to a review board that the patient is unresponsive to any other form of treatment.
Speech and other unilateral functions
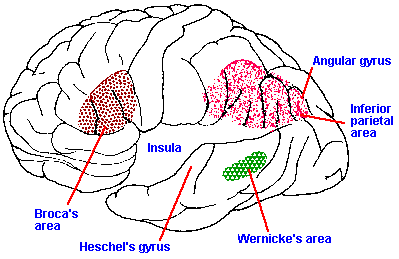 |
Fig. 17-2. A drawing of the lateral aspect of the dominant hemisphere of the human brain showing the approximate locations of Broca's, Wernicke's, and the inferior parietal speech areas. Note that the temporal lobe has been pulled out, exposing the insula beneath. (Geschwind N: Sci Amer 226(4):76-83, 1972)
|
Thus far, everything that has been said about the
nervous system applies equally to both sides, that is, the
nervous system is bilaterally symmetrical. However,
there are some cortical functions that are not served
equally by both hemispheres. In one cerebral
hemisphere, there is a region, called Broca's area, that
is involved in speech (Fig. 17-2, lateral aspect of the
dominant hemisphere). Broca's area (areas 44 and 45)
is located in the frontal cortex just anterior to the face
area of the precentral gyrus. Actually, there are two
additional areas of the same hemisphere that are
involved with speech. These are the inferior parietal
lobule, consisting of the angular and supramarginal gyri
(areas 39 and 40), and Wernicke's area (area 22), located on the posterior aspect of the superior
temporal gyrus just behind the auditory cortex. The location of these three areas in seen in Figure
17-2. These areas are found in only one hemisphere, and their presence defines the dominant
hemisphere. The dominant hemisphere is usually the one opposite the favored hand (in most
individuals, the left hemisphere), but, in a few cases, it is the hemisphere on the same side as the
favored hand (92% of right-handed people are left hemisphere dominant, 50 to 60% of left-handed people are right hemisphere dominant).
Electrical stimulation during neurosurgery in any of these three areas leads to arrest of speech,
hesitation or slurring of speech, distortion, repetition, inability to name, misnaming, and
vocalization. Vocalization can also be elicited from the motor cortex where it always takes the
form of a sustained or interrupted cry.
Various types of speech disorders result from damage to the speech areas. The general term
for these disorders is aphasia. Lesions involving Broca's area and adjacent cortex produce a
motor or expressive aphasia, in which the patient does not or cannot speak. What words are
used are uttered slowly and with great effort. Usually, difficulties also show up in writing words.
In expressive aphasia, it appears that the patient has lost his motor programs for making correct
and properly timed sequences of movements of lips and tongue, although he apparently still
knows what to say. A lesion of Wernicke's area or the inferior parietal lobule produces a
receptive aphasia in which the patient cannot understand spoken language or written language
(alexia), but is capable of spontaneous speech. Many times, lesions of the inferior parietal lobes
of the dominant hemisphere also produce what is known as Gerstmann's syndrome. This
syndrome may include one or more of the following symptoms: dysgraphia, a deficit in writing
without motor or sensory deficits in the upper extremities; dyscalculia, a deficit in the
performance of calculations; left-right confusion; and finger agnosia, errors in finger
recognition. It is seldom that aphasias present themselves simply as described above. Usually, a
given aphasia is a mixture of expressive and receptive difficulties. A number of classification
schemes have been invented, but no one of them can handle all cases. The following case history
illustrates aphasia:
| A 19-year-old soldier was struck in the head at the left temple by a piece of shrapnel. He remained
conscious and remembered everything that happened. When admitted to the hospital 4 days later, he was
speechless. Shortly thereafter he began to make articulate noises that seemed not to be related to words. For
instance, he would utter "ho-nus" when asked his name or "ong" when shaking his head in the negative. He
could understand and follow simple commands, but was slow to follow two or three consecutive orders
(complete inability to follow commands is apraxia). As he improved he was able to say the alphabet and days
of the week, but pronounced the words and letters poorly. He could write his own name and address, but not
that of his mother. He could not count aloud, but could write the numbers up to 21. When asked to write a
simple phrase of dictation, he frequently failed to finish the phrase. Seven months after the injury he still had
great difficulty in evoking appropriate words during spontaneous speech. Pronunciation was poor; writing was
done only with difficulty. Seven years later he still hesitated in searching for appropriate words and showed
defective enunciation and influency in writing. (Head H: Aphasia and Kindred Disorders of Speech.
Cambridge, University Press, 1926) |
The devastating losses in language abilities resulting from even small lesions of speech centers
of the dominant hemisphere suggest that a dominant hemispherectomy should abolish language
altogether. Surgeons were surprised to find that this procedure does not result in complete
aphasia, but in some verbal loss, especially in responses to written commands. Deficits are fewer
in writing than in speech and least in responses to verbal commands. Speech abilities are
recovered to a great extent over a period of 8 months. Why losses due to hemispherectomy are
smaller than predicted is one of the enigmas of neuroscience, yet it should be recalled that the
influence of hemispherectomy on movement is also less than expected.
In a few cases of intractable seizures, physicians have elected to section the corpus callosum
by itself or in combination with the anterior commissure and the hippocampal commissure in order
to stop the spread of the seizures from one hemisphere to the other. In many cases, this
procedure has been successful, and it has given the scientist an opportunity to study the abilities of
each hemisphere in isolation. The separation of the two hemispheres creates, in some senses, two
minds within the same body. The patient, sometimes called a split-brain patient, is able to get
around well and can do most, if not all, of the things he did before, but some things he does in a
subtly different way. For example, in performing tasks that require the cooperation of both hands,
the patient may be severely impaired. If asked to match a pile of blocks by building a new pile
next to it, the patient finds one hand putting a block in place, while the other one takes it away or
moves it somewhere else. He can only do the task successfully if he sits on one hand, letting the
other one do the work. Split-brain patients show no impairment in intellectual functioning. In
fact, in the one case where adequate pre- and postoperative testing was performed, the patient
showed improvement in standardized memory tests, short-term memory, and learning rate.
Apparently, separating the hemispheres does not disrupt these functions. Why they improved in
this one case is a matter of conjecture, and whether this is a consistent result of the surgery awaits
further tests.
Sectioning the corpus callosum surgically has some rather interesting effects on sensation in
the modalities of somesthesia, audition, and vision. The ability to localize the site of a stimulus to
the skin is not impaired in split-brain patients, but if the testing procedure is set up in such a way
that each hemisphere is tested separately, then it can be shown that a hemisphere can only localize
stimuli on the contralateral body surface. Thermal sensitivity and position sense of a hemisphere
are likewise limited to the contralateral side of the body. Complex discriminations of solid shapes
with no limit to the number of alternatives are performed only by the contralateral hemisphere, but
if the alternatives are easily discriminated (as tested using the contralateral hemisphere) and few in
number (e.g., cubes versus spheres), then the ipsilateral hemisphere can discriminate them as well.
It is not known by what pathways the sensory information reaches the ipsilateral hemisphere in the
split-brain patient. When the discriminations are learned by one hemisphere only, the other
hemisphere shows no indication that it "knows" of the learning, that is, there is no intermanual
transfer of tactile discriminations.
There is no loss of auditory sensitivity after the brain is split, and the ability to localize the
source of a sound is unimpaired. The patient can also respond properly, using either hand, to a
verbal command directed at either ear. He cannot, however, fuse two sounds, one presented to
each ear. A normal person, presented with "pro" to the right ear and "duct" to the left ear in
quick succession, hears "product." The split-brain patient hears "pro" with the left hemisphere
and "duct" with the right. The left hemisphere-dominant-patient's verbal report will be "pro" only,
but he will write "duct" using his left hand.
Under conditions where the brain has been surgically split, it is possible, provided that the
eyes do not move, to direct visual stimuli to each hemisphere independently because of the pattern
of crossing of optic tract fibers in the optic chiasm. Objects in the right visual field are seen by the
left hemisphere; those in the left visual field are seen by the right hemisphere; and the macula is
not represented bilaterally. Following the commissurotomy, objects in the right visual field of a
patient with left hemisphere dominant can be correctly named by the patient, those in the left
cannot, presumably because of the location of speech areas. On the other hand, a photograph
shown to either hemisphere can be correctly pointed out using the contralateral hand. The patient
is unable to make same-different discriminations between photographs presented one to each
hemisphere.
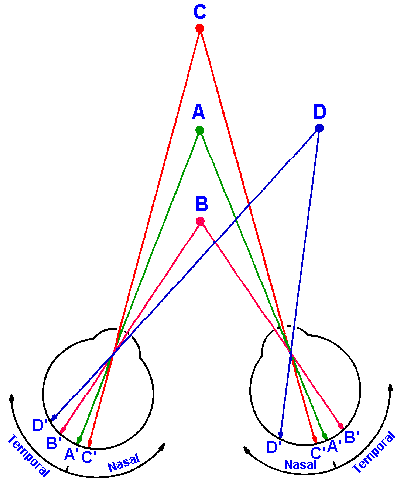 |
Fig. 17-3. The images of an object immediately in front of (B) or behind (C) the fixation point (A) fall on both temporal retinae (B') or both nasal retinae (C') and therefore are transmitted to different hemispheres. The image of an object anywhere else (D) falls on one nasal and one temporal retina (D') and therefore is transmitted to only one hemisphere.
|
Split-brain patients have difficulty with stereopsis
(depth perception) when an object lies immediately in
front of or behind the fixation point. This is because the
images fall on the retina so that they are transmitted to
different hemispheres, and determining whether to
converge or diverge the eyes is impossible. In Figure 17-3, the eyes are fixated on point A; the images of points B
and C fall on the temporal retinae and nasal retinae,
respectively, and therefore are transmitted one to each
hemisphere. So in the case of either point B or C, neither
hemisphere would have both images to compare, the
basis upon which decisions about convergence and
divergence are normally made. When both images of an
object fall on the retinae so that they are transmitted to
the same hemisphere (as in the case of point D in Fig. 17-3), there is no problem.
The question arises whether the nondominant
hemisphere has any verbal capabilities at all. To test this,
a word was flashed in the opposite visual field, and the patient was asked to pick the word out of
a number of others. The nondominant hemisphere can do this, at least for some kinds of words.
It has no difficulty in recognizing nouns and adjectives. Apparently, the nondominant hemisphere
does have language skills, but these are not manifested normally, because it has no access to the
speech apparatus. They are clearly seen, however, if written responses are required.
The nondominant hemisphere appears to be better at visual form perception than the dominant
one, at least for complex forms and faces. This is shown by the following tests. Split-brain
patients were asked to copy line drawings that suggested spatial perspective, for example, a cube.
Performance was consistently better with the left hand than with the right hand despite the fact
that all patients were right handed and had little experience drawing with their left hands.
Evidence indicates that the left hand receives its major motor control from the right, in these
cases, nondominant hemisphere. Assembling complex puzzles is also much easier for these
patients when they use their left hands.
This superiority of the nondominant hemisphere for form perception also shows up when no
fine motor coordination is required by the test. Two photographs, each of a different face, are cut
along the vertical midline of the face. The left half of one face is put together with the right half
of the other, forming a composite face. If a split-brain patient fixes his gaze on the junction
between the two half-faces, the left half-face is seen by the right hemisphere and the right half-face
is seen by the left hemisphere. If the composite face is flashed briefly so that the eyes will not
have time to move, then neither hemisphere is aware of the half-face in the ipsilateral visual field.
The patient, asked to point out which of the original faces he saw in the brief flash, nearly always
selects the one, half of which was seen by the right, nondominant hemisphere; if asked to describe
the face he saw, he describes the face, half of which was seen by the dominant hemisphere and
never reports seeing two halves of different faces.
In spite of what was earlier said about lesions of the inferior parietal lobe of the dominant
hemisphere (Gerstmann's syndrome), the calculating ability of the nondominant hemisphere
appears to be superior to that of the dominant one. The test is to give the right and left
hemispheres addition and subtraction problems and require an answer to be selected from among
several alternatives by pointing, the tests not requiring the use of language. Problems can be
presented to only one hemisphere at a time by flashing them in only the left or right visual field
when the eyes are not moving. The superiority of the nondominant hemisphere, shown in this test
in terms of number of correct responses, does not show up in tests requiring verbal responses.
This is presumably the reason for the dyscalculia of dominant hemisphere lesions; the ability is
normally tested using verbal responses.
The nondominant hemisphere appears to be better at some sorts of tests of creativity involving
making associations between concepts, similar to the Miller Analogies Test you may have taken.
Still other aspects of creativity seem to be the province of the dominant hemisphere.
Apparently, each hemisphere has a sense of self, recognizing the individual's name and
supplying it when requested. Each hemisphere has its own system of subjectively evaluating
current events, planning for future events, setting response priorities, and generating personal
responses. Both hemispheres are capable of the same emotional responses. A picture of a nude
person of the opposite sex evokes the same emotional response whether it is shown to the
dominant or nondominant hemisphere of a split-brain patient. The emotional responses and
evaluations of people and things by the two hemispheres can also come into opposition with each
other. There are reports of a man who shook his wife with the left hand and came to her rescue
with the right and of a man who pulled down his pants with one hand and pulled them up with the
other.
In split-brain patients, it has been observed that when the two hemispheres agree on
evaluations of people and other matters, the patients outwardly appear calm and tractable, but
when the two hemispheres disagree, they are outwardly hyperactive, agitated, and aggressive. It
would appear that the two hemispheres are also capable of reading differences in each other. In
one experiment, words were flashed tachistoscopically to one hemisphere or the other, and the
patient, in this case a 15-year-old boy, was asked to "do what the word says." When the word
"kiss" was flashed to the right (mute) hemisphere, the patient immediately responded, "Hey, no
way, you've got to be kidding." When asked what the word was he responded, "nurse." When
"kiss" was flashed to the left (verbal) hemisphere, he responded in the same tone, "No way. I'm
not going to kiss you guys." How the verbal hemisphere could read the emotional content of the
word in both exposures is unknown, but clearly it did (Gazzaniga MS, LeDoux JE, Wilson DH:
Neurol 27:1144-1147, 1977).
The dominant hemisphere clearly has the major role in complex motor functions as indicated
by the existence of a preferred hand, but the nondominant hemisphere is perfectly capable of
controlling its half of the body on its own. There is even some indication of ipsilateral control of
muscles other than finger muscles; however, it is not known to what extent this control is the
result of cross-cuing, that is, using small compensatory changes in the midline postural muscles as
indicators of motor performance, and to what extent there is direct control by sensory feedback to
the ipsilateral hemisphere. In some surgical cases, an entire hemisphere has been removed down
to the thalamus, and in these cases both sides of the body move about equally well. The two
hemispheres do compete with each other in split-brain patients. Such a patient can easily build a
simple jigsaw puzzle with either hand, but has great difficulty using both hands, because as one
hand puts the pieces in place, the other removes them. Recall that complex puzzles are only
constructed with ease by the hand contralateral to the nondominant hemisphere because of its
spatial abilities. The two hemispheres normally work together, communicating with each other by
way of the commissures, to produce normal behavior, both in terms of movement and sensation
and in terms of covert processes like calculating and reasoning. It is only when one hemisphere is
not functioning or when the two are functioning independently that we can see that they are not
equal. There are a number of known cases of callosal agenesis, cases in which the corpus
callosum fails to develop. The same tests used to study split-brain patients have also been applied
to patients with callosal agenesis. They have little deficit in visual or tactile tests of the sorts
already described, but they do have deficits in certain spatiomotor tests, e.g., tactile maze
learning, regardless of whether the tests required interhemispheric transmission. They also had
difficulty with certain purely motor tasks, yet the investigators concluded that they have "fewer
disconnection symptoms" than split-brain patients. It is possible that use of other undamaged
commissures may explain the differences between the results of agenesis and surgical section.
The differences should not be entirely surprising; the results of damage to the brain early in life are
often different from those of damage occurring in adults.
It is not known how this asymmetry in the cerebral hemisphere comes about. Some
investigators believe that the dominance of one hemisphere develops as a result of experience in
early life. One theory links the development of language skills to reinforcement of handedness by
parents, but the existence of left dominant, left-handed people seems to contradict this theory.
Other investigators maintain that the asymmetry is part of the heredity of the individual and that it
merely shows itself gradually as behavior develops. Clearly, there are some asymmetries in
handedness and in neural responses to speech sounds in infants. These would appear to be innate.
Yet, some verbal asymmetries have not been found in boys until 5 to 7 years of age and in girls
until 9 to 11 years of age. Whether this late appearance is innate or learned, the difference in time
of onset in the sexes may help to explain the greater resistance of girls to permanent damage to
language function caused by early brain trauma. It may be that either hemisphere can become
dominant, but only one does under normal circumstances.
There are clear structural differences between the two hemispheres that may underlie these
differences in hemispheric functions. There are, of course, the larger "speech areas" in the
dominant hemisphere. In addition, auditory primary sensory and association areas appear to be
larger in the dominant hemisphere than in the nondominant, whereas the temporoparietal cortex
appears to be larger in the nondominant hemisphere. In right-handed people, most of whom are
left hemisphere dominant, the frontal lobes are more often wider in the right and the occipital
lobes more often wider in the left hemisphere. There are many more structural asymmetries in the
brain, but we do not know which may be associated with dominance. Structural asymmetries
have also been found in fossil human and protohuman brains, in all of the living great apes, and in
some of the lesser apes and monkeys. Again, the relationship of these asymmetries to language is
unknown; in fact, we are only just becoming aware of the extent of language capabilities in
animals.
Such asymmetries are not unique to the brains of mammals. Birds also show asymmetries in
areas associated with the production of song, and there is a clear hemispheric dominance in the
control of song. The lateral asymmetry in birds is maintained down to the motoneurons that
innervate the syrinx. This dominance is interesting in light of the fact that the bird's brain is so
different in structure from the mammalian brain.
Summary
Consciousness, in terms of the state of arousal, is determined by subcortical structures,
whereas the content may be more the province of the cortex. Sleep is controlled by brain-stem,
hypothalamic and subthalamic mechanisms, but the cortex may also play a role in its regulation.
Emotional behaviors, including both sensory and motor aspects, seem to be a function of the
limbic system. Learning is a property of the entire nervous system, but instrumental or operant
conditioning and discrimination learning can be severely impaired by cortical lesions. Memory
consists of both short-term and long-term events. Most damage to memory involves interferences
with conversion of short-term to long-term memory, a function that involves the hippocampus.
Memory traces may be stored bilaterally, and they can be stimulated during brain surgery causing
them to be played back. Personality is determined, at least to some extent, by the prefrontal
lobes. Lobotomies produce asocial behavior, distractibility, perseveration, inability to plan ahead
and inability to postpone gratification. The two cerebral hemispheres are not equivalent. One is
dominant by virtue of its possession of speech centers in Broca's, Wernicke's, and the inferior
parietal areas. The nondominant hemisphere, is superior in calculation and recognition of spatial
relations. Each hemisphere controls movements of and receives sensory information
predominantly from the opposite side of the body. It normally shares its knowledge and control
by way of the commissures.
Suggested Reading
- Burkland CW, Smith A: Language and the cerebral hemisphere. Neurology 27:627-633,
1977.
- Chiarello C: A house divided? Cognitive functioning with callosal agenesis. Brain and
Language 11:128-158, 1980.
- Curtis BA, Jacobson S, Marcus EM: Introduction to the Neurosciences. Philadelphia, WB
Saunders, 1972.
- DeJong RN, Itaboshi HH, Olsen JR: Memory loss due to hippocampal lesions. Arch Neurol 20:339-348, 1969.
- Gazzaniga MS: The Bisected Brain. New York, Appleton-Century-Crofts, 1970.
- Gazzaniga MS, LeDoux JE, Wilson DH: Language, praxis, and the right hemisphere: Clues
to some mechanisms of consciousness. Neurology 27: 1144-1147, 1977.
- Geschwind N: Specializations of the human brain. Sci Amer 241:180-199, 1979.
- Glickstein M: Neurophysiology of learning and memory. In Ruch TC, Patton HD [eds]:
Physiology and Biophysics. I. The Brain and Neural Function, 20th ed. Philadelphia, WB
Saunders, 1979.
- Head H: Aphasia and Kindred Disorders of Speech. Cambridge, University Press, 1926.
- Kluver H, Bucy PC: "Psychic blindness" and other symptoms following bilateral temporal
lobotomy in Rhesus monkeys. Amer J Physiol 119:352-353, 1937.
- Olds J, Milner P: Positive reinforcement produced by electrical stimulation of septal area and
other regions of rat brain. J Comp Physiol Psychol 47:419-427, 1954.
- Papez JW: A proposed mechanism of emotion. Arch Neurol Psychiat 38:725-743, 1937.
- Penfield W: The neurophysiological basis of thought. Mod Perspectives Psychiat 1:313-349, 1971.
- Rosenzweig MR, Bennett EL [eds]: Neural Mechanisms of Learning and Memory.
Cambridge, MA, M.I.T. Press, 1976.
- Scoville WB, Milner B: Loss of recent memory after bilateral hippocampal lesions. J Neurol
Neurosurg Psychiat 20:11-12, 1957.
- Tsukada Y, Agranoff BW [eds]: Neurobiological Basis of Learning and Memory. New
York, Wiley, 1980.
- Wyler AR, Burchiel KJ, Robbins CA: Operant control of precentral neurons in monkeys:
Evidence against open loop control. Brain Res 171:29-39, 1979.
[TOC] [Chapter 18] [Glossary] [Index] [Abbreviations]
|
 Chapter 17
Chapter 17