VESTIBULAR SYSTEM
The vestibular apparatus is the nonauditory portion of the inner ear. It serves three primary purposes in man: (1) it plays the dominant role in the subjective sensation of motion and spatial orientation of the head, (2) it adjusts muscular activity and body position to maintain posture, and (3) it stabilizes in space the fixation point of the eyes when the head moves, providing a stable image upon the retina. It is important to keep in mind that the vestibular apparatus provides to the senses and to these other control systems information about the position of the head in space. These sensors can detect a change in orientation of the head of 0.5° from the upright position, a change of 5° from the horizontal position, or a change of 15° from the upside down position.
Because the vestibular apparatus provides this information only with respect to the head, it cannot accomplish postural adjustments on its own. Sensors in the neck and perhaps other postural muscles are extremely important in signaling to the central nervous system changes in the relationship between the head and the body. These two systems function in conjunction with the eyes to form a remarkable control system that maintains us in an erect position in a wide variety of stable and unstable postures. The importance of neck proprioceptors in controlling posture is illustrated in certain injuries incurred in automobile accidents. Whiplash injuries can lead to signs of vestibular malfunction, not because they damage the vestibular apparatus but because they interfere with the functioning of proprioceptors in the neck muscles. With these injuries, the central nervous system apparently knows where the head is, but cannot get an accurate position for the body.
This control system can function adequately in certain situations without the vestibular input. People with labyrinthine disease or damage to the vestibular nerve, as sometimes occurs with inappropriate streptomycin therapy, can still orient in space, provided they keep their eyes open or maintain a very rigid relationship between head and body. It is a difficult activity, requiring considerable concentration, and is not the automatic activity that the vestibular apparatus provides.
| Vestibular anatomy |
The basic design of the labyrinthine organs was discussed in Chapter 8 with respect to the auditory structures. The vestibular apparatus consists of the same tube (membranous labyrinth) within a tube (bony labyrinth) construction. Both tubes are fluid-filled: endolymph in the membranous and perilymph in the bony labyrinth. The vestibular apparatus contains two sack-like structures, the utricle and saccule, and three semicircular canals that are joined together in the same fluid environment. The utricle and saccule are termed otolith organs because they contain some crystals of calcium carbonate that play a major role in their excitation. Both otolith organs are dilatations of the labyrinth that contain sensory epithelia, specialized to sense static position and linear acceleration.
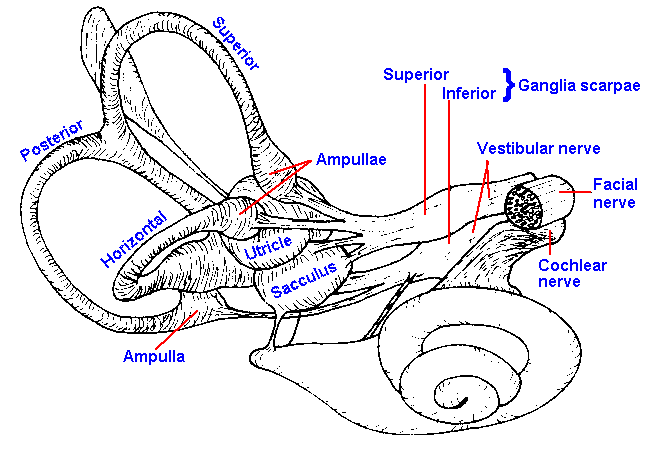 |
Fig. 9-1. A nearly lateral view of the right human labyrinth. (Hardy M: Anat Rec 59:403-418) |
The utricle is an oval tube, whereas the saccule is flattened and irregular; otherwise, their structures are similar. Their positions within the inner ear are shown in the drawing of Figure 9-1. The sensory epithelium of the utricle, the utricular macula, is situated so that it is approximately parallel to the ground when the head is upright. The saccular macula is oriented vertically in the upright human. Both macular organs, in fact the entire vestibular epithelium, contains two types of sensory hair cells, positioned only on the superficial aspect of the epithelium, as shown in Figure 9-2. The type I cell is goblet-shaped and reminiscent of the sensory cells of the basilar membrane, whereas the type II cell is essentially cylindrical. Projecting from the free surface of each type is one kinocilium flanking a group of stereocilia. This arrangement makes the cells polarized, and it has important functional implications.
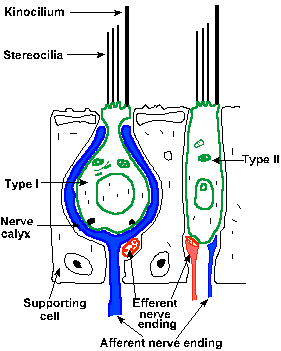
|
Fig. 9-2. Schematic drawing of the vestibular epithelium showing the two cell types and the nerve connections made on each. (Brodal A: Neurological Anatomy in Relation to Clinical Medicine, 2nd ed. New York, Oxford Univ. Press, 1969) |
The sensory epithelia are overlaid by a gelatinous substance that contains the calcium carbonate crystals and in which the cilia are embedded. These crystals are called the otoliths ("stones in the ear") or statoconia, and the gelatinous membrane that contains them is the otolithic membrane. A schematic diagram of the relationship between the hair cells, the otolithic membrane and gravitational force is shown in Figure 9-3 for three different head orientations.
There are both afferent and efferent fibers to the maculae. Both lose their myelin as they pass through the basement membrane, and each larger afferent fiber contacts a small group of type I sensory cells all with the same polarity, i.e., they all have the kinocilium on the same side of the cell. The afferent fibers form synapses on the sensory cells that nearly completely engulf the basal end of the cell. The smaller afferent fibers contact both types of sensory cells, with different polarities, forming the same type of synapses on type I cells as larger fibers and ordinary boutons (see Chapter 13) on type II cells.
In each ear there are three semicircular canals oriented approximately at right angles to each other, as shown in Figure 9-1. One canal (the horizontal) is located in a plane that is about 30° from horizontal; it can be brought into the horizontal plane by tilting the head forward by 30°. The other two canals (the posterior and superior) are in vertical planes. It is important to remember that they occur on both sides of the head, i.e., there are two horizontal, two posterior, and two superior canals. The two horizontal canals lie in the same plane whereas the posterior canal on one side of the head is in a plane parallel to that containing the superior canal on the other side.
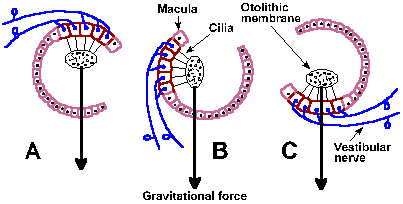
|
Fig. 9-3. The effects of different positions of the head upon the otolith organs. A. Upside down, C. right-side up, and B. in between. The arrows indicate the direction of gravitational force. (Eyzguirre C, Fidone SJ: Physiology of the Nervous System. Chicago, Year Book Medical Publishers, 1975) |
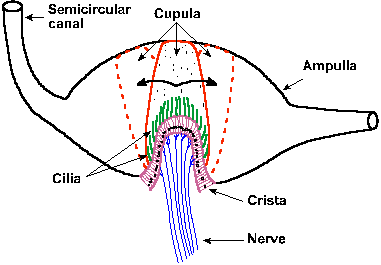 |
Fig. 9-4. The position of the crista ampullaris and cupula within a cross section of the ampulla of one semicircular canal. Also shown is the movement of the cupula and its embedded cilia during rotation first in one direction and then in the opposite direction. (Eyzguirre C, Fidone SJ: Physiology of the Nervous System. Chicago, Year Book Medical Publishers, 1975) |
The lumen of each semicircular canal is connected with the utricle, but the superior and posterior canals join each other before entering the utricle; thus, there are five openings into the utricle (two from the horizontal canal, one each from the posterior and superior canals and one for the joined posterior and superior canals). Near the junction of one arm of each canal with the utricle, there is a dilatation of the canal, called the ampulla. Inside the ampulla and running at right angles across it is the sensory epithelium of the semicircular canal, the crista ampullaris or crista acustica. The hair cells of each crista are similar to those in the maculae except they are all polarized in the same direction. In Figure 9-4, it can be seen that the crista is a folded structure with the cilia of the hair cells projecting out into a gelatinous substance, the cupula. The cupula extends from the crista across the ampulla, dividing it completely and not allowing fluid to pass by it. The result is that if there is a relative movement of the fluid with respect to the canal, the cupula and thus the cilia will be bent to the side.
| Vestibular physiology |
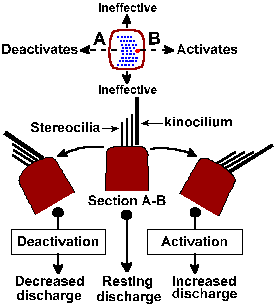
|
Fig. 9-5. Shearing force in vestibular organs. Upper diagram shows arrangement of cilia on a hair cell as seen from above; position of kinocilium indicated by larger dot. Dashed line indicates direction of effective shearing forces; forces at right angles are ineffective. Lower diagram shows section through hair cell along dashed line (upper diagram) with cilia at rest (center) and tilted right and left. Tilt toward kinocilium excites, tilt away deceases excitation. (Klinke R: Physiology of hearing. In Schmidt RF [ed]: Fundamentals of Sensory Physiology. New York, Springer-Verlag, 1978) |
Presumably, the sequence of events in the excitation of vestibular nerve fibers is similar to that for auditory nerve fibers. Movement of the cilia to one side results in a hypopolarization of the hair cell, a receptor potential that in turn releases a transmitter substance that finally hypopolarizes (the generator potential) the afferent fibers that contact it. Thus, when the head tilts forward the gravitational force on the otolithic membrane of the otolith organs changes and the hairs are subjected to a new force that displaces cilia to the side (Fig. 9-3B); pulls them down, away from the epithelium in the inverted position (Fig. 9-3A); or pushes them back against the cell in the upright position (Fig. 9-3C). Maximal discharge of the nerve occurs when the cilia are bent to one side (B); minimal discharge occurs when they are bent to the other side. An intermediate response occurs in any intermediate position (A and C). Because the hair cells are polarized in different directions in various regions of the macula, some hair cells will be excited by movement of the head in any direction. The fibers innervating the utricular macula are slowly adapting and therefore can signal position of the head. They also signal linear acceleration(1) because the forces exerted on the otolithic membrane by such acceleration also cause bending of the cilia, much as a straightened wire coat hanger with a weight on the end is bent when you begin to run with it. The exact function of the saccule is not known.
The semicircular canals are positioned and structured to sense angular rotation. Forces that bend the cilia of hair cells in the cristae are generated by relative movements of the endolymph and the labyrinth. The principle is the same as that which operates in a glass of fluid containing ice cubes. Did you ever try to get a particular ice cube out of such a container? You rotate the glass, but the ice cube stays in the same place with respect to you! The inertia of the fluid makes it lag behind the glass in rotation. However, if you continue to rotate the glass at constant velocity, the fluid and the ice cubes will soon be rotating at the same speed as the glass. Now, if you suddenly stop the rotation, the fluid and the ice will continue to rotate until they gradually slow down and stop. If the ice stops on your side of the glass you get it, if not try again.
When the head is rotated the canals move with it, but the inertia of the endolymph causes it to lag behind. This results in a bending of the cupula in the direction opposite the rotation (dashed outline in Fig. 9-4) and either an increase or a decrease in the discharge rate of the nerve cells innervating the crista, depending upon the direction of movement. If the rotation continues, the endolymph catches up with the movement of the canal; the cupula is returned to the upright position; and the discharge of the nerve fibers returns to its former rate (it adapts). If the rotation is suddenly stopped, the canals immediately stop their rotation, but the endolymph does not, so the cupulais bent in the other direction. The effect on the discharge in the vestibular nerve fibers is opposite that of the original rotation.
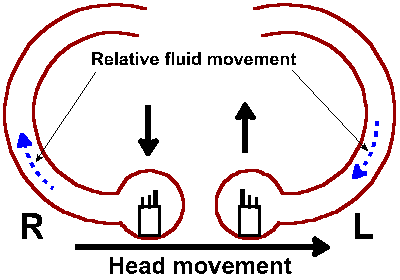 |
Fig. 9-6. Schematic of the horizontal semicircular canals complete with hair cells sporting stereo- and kinocilia. Direction of a leftward head rotation is indicate as are the relative fluid movements in the canals. The effect of the fluid movement on the two hair cells is indicated by the bold upward and downward arrows. |
Because of the nearly orthogonal orientations of the semicircular canals, rotation in any direction will be sensed by one or more of the cristae. As in any Cartesian three coordinate system, any direction of rotation can be signaled uniquely by the combined discharges from the three canals. In the horizontal canal, the kinocilium is polarized toward the utricle. The upper part of Figure 9-5 shows the arrangement of the cilia on a hair cell and the effects of movements of the cilia in different directions. The lower part shows that when the cilia are bent in the direction of the kinocilium, the vestibular nerve fibers are excited, and when they are bent the other way, the discharge is reduced. This means that in the horizontal canals, the vestibular nerve activity increases when the endolymph moves (relatively) in the direction of the ampulla. Thus, when the head rotates to the left, the fluid in the right horizontal canal moves away from the ampulla (reduced excitation), and the fluid in the left one moves toward it (more excitation). This is shown graphically in Fig. 9-6. The reverse movement or a deceleration has the opposite effects. In the vertical canals, the hair cells are polarized away from the utricle. Thus, the activity originating in the right superior canal is increased and that from the left posterior canal is reduced by rotating the head forward and to the right, whereas the reverse effects are seen when the head is rotated backward and to the left. With the head tilted forward by 30°, the horizontal canals are activated exclusively by movement around the vertical axis (the movement is like a "no" gesture with the head); the posterior canal on one side and the superior canal in the other labyrinth are activated by movement of the head in the anterolateral or posterolateral direction (like a "yes" gesture but at a bit of an angle). These can be seen by reference to Fig. 9-7. Note that effects are always bilateral and reciprocal. That is, when one horizontal, posterior or superior canal is excited, the contralateral horizontal, superior or posterior canals has its activity reduced.
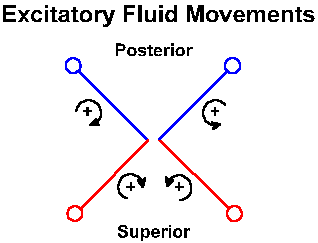
|
Fig. 9-7. Schematic of the two superior and two posterior semicircular canals. Curved arrows are meant to give a 3-d representation of the relative fluid movement within each canal that excites that canal. Of course, the head movement that produces the indicated fluid movement would be in the direction opposite that indicated by the arrow. |
| Electrophysiology of the vestibular nerve |
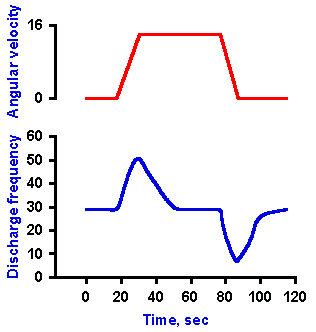 |
Fig. 9-8. Response of semicircular canals to angular rotation. A. The velocity of an angular rotation of the head (ordinate) plotted against time (abscissa). B. The change in frequency of discharge of a receptor innervating one of the semicircular canals during periods of constant acceleration, of constant velocity, and of constant deceleration of the head. |
Recordings made from single axons innervating the monkey's semicircular canals indicate that they have a high resting discharge rate and increase their discharge rates with angular acceleration; however, the receptors adapt rapidly in response to constant angular velocity, adapting completely to resting rates in less than one minute. The neurons decrease their discharge rates with angular deceleration, and adaptation also occurs with constant (decreased) angular velocity. This is indicated by the records in Figure 9-8. The upper record shows the angular velocity of the head, starting at zero, increasing at a constant acceleration to some greater value, remaining constant at that value for a time before returning at constant deceleration to zero velocity. The discharge rate of the neuron, shown in the lower record, increases from resting rate during constant acceleration, adapts with a time constant of 6.7 sec (tA in Fig. 9-9) back to the resting discharge rate while velocity remains constant at the new value. Discharge frequency decreases during constant deceleration and adapts (this time increasing discharge frequency) to the resting rate with a time constant of 4.9 sec (tD in Fig. 9-9). Figure 9-9 shows a simultaneous plot of responses of a neuron innervating the horizontal semicircular canal to constant accelerations (open circles) and decelerations (filled circles) to illustrate the longer time constant of adaptation to accelerations. All receptors in the same ampulla have the same polarization so all are excited or inhibited by acceleration in the same direction.
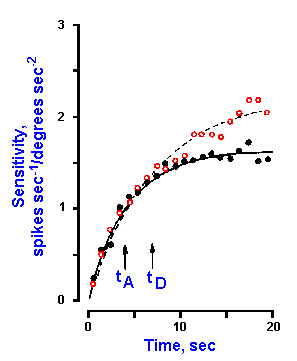 |
Fig. 9-9. Time course of responses to constant acceleration (open circles) and deceleration (filled circles). Sensitivity to acceleration in spikes/sec/degree/sec2 (ordinate) is plotted against time (abscissa). The vertical arrows indicate the acceleration and deceleration time constants, tA and tD, for a slightly adapting neuron from the horizontal semicircular canal. (Goldberg JM, Fernandez C: J Neurophysiol 34:635-660, 1971) |
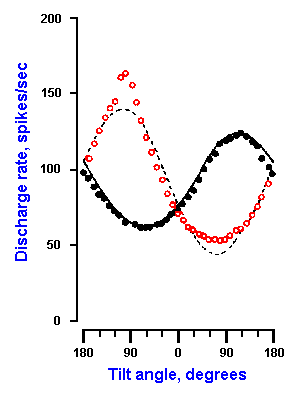 |
Fig. 9-10. Response curves for a utricular receptor to various pitches and rolls. Discharge rate (ordinate) is plotted against tilt angle (abscissa). Positive angles correspond to forward pitch and ipsilateral roll. Open circles are for pitch, filled circles for roll. (Fernandez C, Goldberg JM, Abend K: J Neurophysiol 35:978-997, 1972) |
In contrast, receptor cells in the utricle are not all polarized in the same direction so not all the neurons respond to the same sort of movement. For example, 70% of the neurons in one study were most sensitive to ipsilateral sideways roll of the head, whereas contralateral roll was most effective in exciting 30% of the neurons. About half of the neurons were most sensitive to nose up pitch and half to nose down pitch. An example of the discharge characteristics of a utricular receptor to pitches (open circles) and rolls (filled circles) is shown in Figure 9-10. Positive angles in the figure correspond to forward pitch and ipsilateral roll. This particular receptor increased its discharge rate with nose up (backward) pitch until the angle of pitch reached about 90°; thereafter the discharge decreased. The discharge rate decreased with nose-down (forward) pitch to about 60° and increased with larger forward pitches. The response of the neuron to an ipsilateral roll was an increased discharge up to about 120°, then a decrease, whereas the response to a contralateral roll was a decrease, then an increase.
| Vestibulo-ocular reflex |
The vestibular nerve fibers project to the vestibular nuclei and to the flocculonodular lobe of the cerebellum. The vestibular nuclei give rise both to the vestibulospinal tracts, which play a role in movement and posture, to be discussed in Chapter 16, and to connections with cranial nerve nuclei III, IV, and VI and motoneurons of the neck by way of the medial longitudinal fasciculus (MLF). The various semicircular canals make preferred connections with motoneurons in the cranial nerve nuclei that control particular eye muscles. Horizontal canals drive motoneurons of the abducens nucleus (sixth cranial nerve) that innervate the contralateral lateral rectus muscle and of the oculomotor nucleus (third cranial nerve) that innervate the ipsilateral medial rectus muscle, while producing appropriate inhibition in the antagonist muscles. The vertical canals drive motoneurons that innervate the superior and inferior recti and oblique muscles.
|
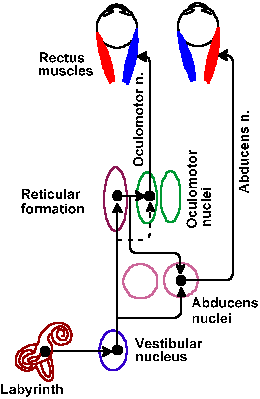
Fig. 9-11. Brainstem pathways for control of eye movements by the left horizontal semicircular canal. (Gruesser O-J, Gruesser-Cornehls V: Physiology of vision. In Schmidt RF [ed]: Fundamentals of Sensory Physiology. New York, Springer-Verlag, 1978) |
One result of these connections is termed the vestibulo-ocular reflex. The neural pathways of the reflex controlled by the left horizontal semicircular canal are shown in Figure 9-11. If the head is moved to the left, the eyes are moved conjugately to the right, in order to maintain the gaze fixed on an object as the head moves. The mechanism for this reflex has been worked out in some detail. As the head is rotated to the left, the discharge of the vestibular fibers supplying the left horizontal canal is increased; concurrently, the discharge on the right is decreased. This results in an excitation of the right abducens nerve fibers innervating the right lateral rectus muscle and the left oculomotor nerve fibers innervating the left medial rectus muscle. The ensuing muscle contractions turn the eye to the right. The reflex, of course, also occurs when the head is rotated in the other direction, but the eye movement is in the reverse direction as well.
The vestibulo-ocular reflex has an important clinical use. As a result of the reflex, when the head is moved from side-to-side, the eyes remain fixed on an object by moving from side-to-side but in the opposite direction. This is called the doll's-eye maneuver by analogy to old-fashioned dolls with counterweighted eyes. It is a normal response to head rotation. Its absence in cases of head injury is usually taken to indicate involvement of the MLF and, therefore, a lesion of the brain stem. There are also reflex rotations of the eyes in compensation for rotation of the head toward the shoulder which involve the vertical canals; these too are normal responses.
| Nystagmus and the caloric test |
The vestibular system, because of its bilaterality, is a balanced system. Effects of activity from one vestibular organ are balanced by effects from the other. A vestibular imbalance will cause overstimulation on one side and slow tonic deviation of the eyes away from that side. Cortical mechanisms rapidly return the eyes to their former position. The tonic nature of the movement induced by the vestibular system causes this slow movement followed by a rapid or saccadic return to be repeated over and over. This complex of movements is called nystagmus. By convention, the direction of the nystagmus is taken to be the direction of the quicker and more easily observed saccadic movement. It must be remembered that this is opposite the movement by vestibular activity. Overstimulation can result in several ways. An irritative lesion can produce overstimulation ipsilaterally; natural stimulation by rotation can produce overstimulation ipsilaterally, but a destructive lesion will produce overstimulation contralaterally.
The presence of nystagmus in a patient usually indicates some problem with the labyrinth, vestibular nerve, or vestibular nuclei, but it also occurs as a part of the cerebellar dysfunction following over-consumption of alcohol. Nystagmus also results from certain viral infections involving the vestibular system. In labyrinthine disease, the nystagmus is usually independent of gaze, but with more central lesions it usually occurs in the direction of gaze. Vertical nystagmus is nearly always evidence of a central nervous system lesion.
The functioning of the vestibular system is often checked clinically by means of the caloric test. For this test, the head is tilted backward by about 60 to bring the horizontal canal into a vertical position. Then either warm or cold water is introduced into the external auditory meatus on one side. The immediate results are usually vertigo (sensations of spinning), nausea, and twisting of the head and body. Nystagmus also results. When warm water is introduced into the meatus, it may set up convection currents in the nearby horizontal canal. The warm endolymph would rise toward the ampulla and stimulate the vestibular nerve fibers there. This increase in activity, whether caused by convection currents or some other phenomenon created by the warm water, results in nystagmus toward that side of the head (the vestibular activity deviates the eyes in the other direction). Cold water induces nystagmus toward the opposite side of the head, apparently by reducing activity in the vestibular nerve (remember: a balanced system). With diseases involving the cerebral cortex, the saccadic phase of nystagmus is sometimes absent, and the eyes merely deviate to a side appropriate for the temperature of the water.
| Responses to angular rotation |
The responses of individuals to rotation have been studied quite thoroughly because understanding them is important to controlling performance in high speed aircraft or in spacecraft. When a person, with eyes closed, is submitted to angular rotation, for example in a Barany chair, he will accurately signal the direction of rotation when he first begins to move; however, after a period of rotation at constant velocity, he will report that he has ceased to rotate. This is precisely what is predicted from the rapid adaptation of receptors in the semicircular canals at constant velocity (Figs. 9-6 and 9-7). During the period of acceleration at the beginning of the rotation, the person will also experience a nystagmus in the direction of rotation, the eyes apparently attempting to stay fixated on some target. When the sensation of rotation fades at constant velocity, the nystagmus also disappears.
If the chair is abruptly stopped at this point, the person will have a sensation of rotation in the direction opposite to that he previously experienced, and that too will fade with time. In addition, there will be a postrotatory nystagmus also in the direction opposite to the previous rotation. Again, these are phenomena consistent with the discharge properties of receptors in the semicircular canals. When a rotation stops, the discharge frequency of the canal receptors (on the side toward which the head originally rotated) falls below resting levels and below the level of the contralateral canal, just as it would if there were actually a rotation in the opposite direction. Although rotation has ceased, the central nervous system cannot distinguish this signal from the signal that would occur for an opposite rotation and, therefore, it is interpreted as an opposite rotation. This sensation also fades with time because of receptor adaptation.
Another consequence of rotation in the postrotatory period is past-pointing. If a person is asked to point at a target immediately upon being stopped from a period of constant velocity rotation, he will consistently point inaccurately, with the deviation always in the direction of the previous rotation. Thus, if the previous rotation was clockwise, he will consistently point to the right of the target.
People who must undergo this kind of rotation frequently learn to ignore what their vestibular senses tell them. Figure skaters, for example, can spin for long periods without showing past-pointing or postrotatory nystagmus. Obviously, they must be able to do this if they are to continue skating after the spin. Airplane pilots can suffer what is called a Coriolis effect, a false sense of spinning. A pilot who has put his airplane into a spin, either intentionally or accidentally, may be unaware of it after a while if he relies upon his vestibular sense because of the adaptation of the receptors. When he stops the spin (usually because someone tells him to), he will experience a sensation of spinning in the opposite direction, the Coriolis effect. Were he then to attempt to correct for this illusory spin, he would set himself in a spin again. Many aircraft have been sent spinning to earth by pilots who failed to read their instruments and relied on the Coriolis effect to navigate.
| Disturbances of vestibular function |
The most common disturbance related to vestibular function is motion sickness. This condition is characterized by vertigo, nausea and vomiting and is apparently primarily precipitated by repeated vertical movements. Most people adapt readily to such movements, but particularly sensitive people become ill when traveling in a car, boat, or airplane. Motion sickness often can be avoided by eating sparing, fat-free meals or by taking antihistamines such as meclizine or dimenhydrinate (Dramamine) before traveling.
Vertigo, the false sensation that either the person or his world is rotating, is associated with vestibular malfunction. Clinicians, but not the man on the street, distinguish vertigo from dizziness, which is a disturbed sense of relationship to space or a sensation of unsteadiness with a feeling of movement within the head. Paroxysmal attacks of vertigo may cause the patient to fall down because of his postural responses to the false sensations of movement. Vertigo may occur acutely, as a result of labyrinthitis, or chronically, as a result of damage to the eighth nerve. The symptoms of Meniere's disease, resulting from an accumulation of endolymph within the inner ear, can involve deafness or tinnitus, ringing or humming in the ears, because of the involvement of the cochlea, and also vertigo, because of labyrinthine involvement. The cause of Meniere's disease is unknown. The vertigo component of the disease can be treated surgically by labyrinthectomy or partial section of the eighth nerve, though the latter procedure is now seldom used. Meniere's disease is also treated pharmacologically with low-salt diets and diuretics as well as with antihistamines or histamine (a desensitizing procedure) or with large doses of thiamine. Vertigo can also be caused by eighth nerve tumors or ill-advised, prolonged treatment with streptomycin.
Rapid destruction, as opposed to slow destruction by disease, of a labyrinth leads to short-term disturbances of equilibrium, vertigo, and nystagmus. These are symptoms of a disbalance of the vestibular inputs; recall that the vestibular system compares the signals from the two labyrinths. With time, most of these symptoms weaken and disappear. For example, in cats a unilateral labyrinthectomy produces contralateral nystagmus (overstimulation opposite the lesion), head tilt toward that side, and a tendency for the body to roll toward the lesion. After three days, only the head tilt remains. Bilateral destruction of the labyrinths causes only a transient (lasting months) disturbance in equilibrium, but even this can be compensated by the positional information supplied by the eyes. In the cat again, bilateral lesions do not lead to nystagmus or tilt; however, walking is broad-based and swaying, and there is no righting reflex if the eyes are covered. After two weeks there is considerable improvement. In man, compensation for labyrinth destruction is complete; as long as the eyes are open, no chronic effects are obvious. The importance of the neck reflexes to movement and posture are emphasized by the observation that a bilateral block of the C1 to C3 dorsal roots produces the same kind of effect on an animal as bilateral labyrinthectomy (see Cohen LA: J Neurophysiol 24:1-11, 1961).
| Summary |
The vestibular apparatus signals both orientation relative to gravity and linear acceleration, through activity from the utricle and possibly also the saccule. The semicircular canals detect angular rotation, but they adapt, so the sensations they produce fade. Both the otolith organs and the semicircular canals are inertial systems, i.e., they depend upon the inertia of solid (the otolith organs) or fluid masses (the semicircular canals) for their operation. The vestibulo-ocular reflex is an induced deviation of the eyes away from the direction of rotation of the head and results from vestibular connections to the ocular motoneurons via the MLF. Nystagmus is a complex sequence of slow and fast eye movements in opposite directions, due to vestibular imbalance and cortical resetting activity. The caloric test is used to check vestibular functioning. Disturbances of vestibular function include motion sickness, a failure to adapt to repeated movements; vertigo, a false sense of rotation due to irritation of vestibular nerve fibers; and short-term disturbances of equilibrium associated with destruction of one or both labyrinths. Position of the body in space is signaled by the vestibular system, by the eyes, and by neck proprioceptors. Partial compensation can occur for the loss of any of these three sources of information, but not for loss of all three.
| Suggested Reading |
- Brodal A, Pompeiano O [ed]: Basic Aspects of Central Vestibular Mechanisms. Amsterdam, Elsevier, 1972.
- Cohen LA: Role of eye and neck proprioceptive mechanisms in body orientation and motor coordination. J Neurophysiol 24: 1-11, 1961.
- Flock A: Sensory transduction in hair cells. In Loewenstein WR [ed]: Handbook of Sensory Physiology, Vol. I. Berlin, Springer, 1971.
- Hallpike CS: The caloric test: a review of its principles and practice with especial reference to the phenomenon of directional preponderance. In Wolfson R [ed]: The Vestibular System and its Diseases. Philadelphia, Univ Pennsylvania Press, 1966.
- Kornhuber HH [ed]: Handbook of Sensory Physiology, Vol. VI. Vestibular System, Part l. Basic Mechanisms. Berlin, Springer, 1974.
- Wersall J, Flock A: Physiological aspects of the structure of vestibular end organs. Acta Otolaryngol (Suppl) (Stockholm) 192:85-89, 1964.
- Wilson VJ, Melvill Jones G: Mammalian Vestibular Physiology. New York, Academic Press, 1979.
- Young LR: Role of the vestibular system in posture and movement. In Mountcastle VB [ed]: Medical Physiology, 13th ed. St. Louis, Mosby, 1974.
Footnotes:
1. Remember Einstein showed that gravity and linear acceleration are equivalent!
[TOC] [Chapter 10] [Glossary] [Index] [Abbreviations]