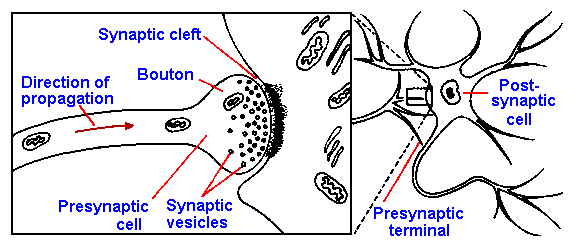
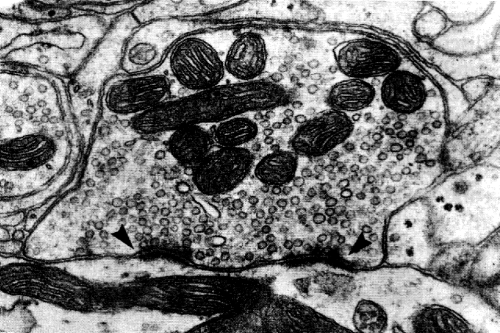
|
Several other types of connections between cells have been discovered. These junctions,
thought to be substrates for chemical transmission, occur between two dendrites
(dendrodendritic synapses), a dendrite and an axon (dendroaxonic synapses), a dendrite and a
soma (dendrosomatic synapses), and any part of a neuron and a node of Ranvier (nodal
synapses). In some cases, synapses have been described in which a single element is both pre-
and postsynaptic to a second element. This arrangement, a reciprocal synaptic arrangement, is
illustrated in Figure 13-2. Note the accumulation of synaptic vesicles on what is presumed to be
the presynaptic side of each synapse. The function of this arrangement is unknown. The
physiology of these new synaptic arrangements has not been studied in detail yet, but it can be
presumed for now that they behave like other chemical synapses. Their presence certainly
broadens the possibilities for interactions between neurons in the central nervous system.
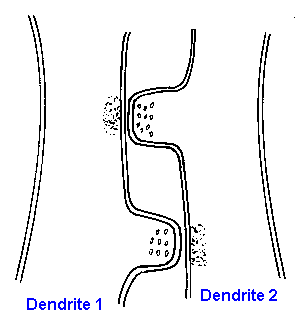 |
Fig. 13-2. A reciprocal synaptic relationship between two dendrites. Note the accumulations of synaptic vesicles on what is presumed to be the presynaptic side of the synapses.
|
Another type of synaptic arrangement that has received a
great deal of attention is the electrotonic synapse or
electrical synapse, the substrate for which is thought to be
the gap junction. A schematic diagram of a gap junction
(sometimes called a connexon) is shown in Figure 13-3. The
membranes of the two cells involved come extremely close or
may actually fuse together. X-ray diffraction studies suggest
that membrane ionic channels (perhaps sodium channels) of
the two cells are in register such that there are small conduits
between the two neurons by which their cytoplasms could
communicate. Gap junctions are most frequently seen in
dendrodendritic, dendrosomatic, and somatosomatic synaptic
arrangements, but they do occur in other types of synapses.
Such junctions have been found in the retina, olfactory bulb,
cerebellar cortex, lateral vestibular nuclei, inferior olive and elsewhere, but they are also found
outside the nervous system. A role for gap junctions in the control of cellular proliferation has
been suggested.
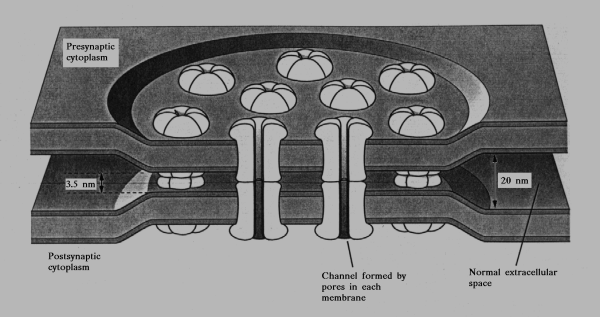 |
Fig. 13-3. A schematic diagram of what is thought to be the structure of an electrical synapse. Note that the sodium channels of the the membranes of the two cells are in register, forming a channel between the two cells.
|

The majority of the synapses in the
vertebrate central nervous system are of the
axosomatic and axodendritic types and of the
chemically transmitting type. It is therefore a
small wonder that chemical transmission is the
most thoroughly understood. There are now
many reports of gap junctions and presumed
electrotonic coupling between neurons in
vertebrates, especially in mammals. Yet, two
important morphological questions still remain
unanswered: (1) Is every structure that looks
like a chemical synapse really a chemical and not an electrical synapse, or for
that matter a synapse at all? (2) Is every gap
junction an electrical synapse and not a chemical synapse, or for that matter a synapse at all?
These questions are made important by the observations that vesicles (possibly synaptic vesicles)
are found in conjunction with both chemical synapses and gap junctions and that gap junctions are
characteristic of epithelia, in general, and not limited to the nervous system, which is epithelial in
origin. It is not clear that answers to these questions are even possible.
| Physiology of a chemical synapse |
An action potential, initiated in an afferent axon, arrives at a bouton and hypopolarizes it.
Boutons hypopolarize in the same way as axons. At this point the action potential itself can go no
farther, but the hypopolarization of the bouton somehow causes some of the synaptic vesicles in
the region to fuse with the presynaptic membrane whereupon the fused membrane breaks and
spills the contents of the vesicles into the synaptic cleft. The release of transmitter substances
depends upon several factors, including the magnitude of the hypopolarization, the number of
available vesicles, and, importantly, the concentration of calcium in the extracellular fluid.
Reduced calcium blocks synaptic transmission. Likewise, increased manganese, a calcium
inhibitor, leads to depression or block of transmission. Calcium appears to be necessary for
hypopolarization-release coupling. Hypopolarization leads to opening of
voltage-gated Ca++ channels and the entry of Ca++ into the terminal. Once inside, Ca++ promotes
fusion of the vesicles with the terminal membrane and release of the transmitter substance. The
released transmitter substance diffuses across the cleft in a fraction of a millisecond and interacts
with the postsynaptic membrane, changing its permeability and, ultimately, the membrane
potential at that point on the postsynaptic membrane. The action of the transmitter substance is terminated by
its removal from receptors on the postsynaptic membrane. Some transmitter substances are then
degraded, e.g., acetylcholine, whereas others are taken up by the presynaptic terminal, e.g.,
norepinephrine and other amine transmitter substances. This entire process, from presynaptic spike to the termination of the postsynaptic
response, frequently requires only 10-20 msec.
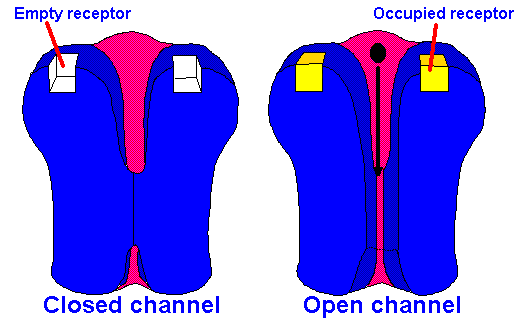 |
Fig. 13-4A. Model of ionotropic synaptic transmission. A transmitter substance binds to a receptor and opens a channel, changing membrane conductance and, therefore, membrane potential.
|
Most chemical transmitter substances act by producing changes in the transmembrane
potential of the postsynaptic cell. Some produce their effects noticeably more slowly than others.
For example, the interval between the arrival of the presynaptic spike at the synapse and the
change in membrane potential, the synaptic delay, is shorter at the neuromuscular junction,
where acetylcholine is the transmitter substance, than at sympathetic postganglionic synapses,
where norepinephrine is the transmitter substance. Surprisingly, acetylcholine acts faster at the
neuromuscular junction than at the parasympathetic junctions at the heart. At ganglionic synapses
of the sympathetic nervous system acetylcholine has both a fast action (4 to 7-msec latency and a
200-msec duration) and a slow action (200 to 300-msec latency and a 5-sec or longer duration).
Both effects occur on the same postsynaptic cell! The rapid-onset, briefer effects of acetylcholine
at skeletal muscle are apparently the consequence of changed ionic conductances, resulting from
conformational changes in the membrane. These conformational changes result from the
interaction of the transmitter substance with receptors(3) in the postsynaptic membrane. This is
called ionotropic transmission, and it is illustrated schematically in Figure 13-4A.
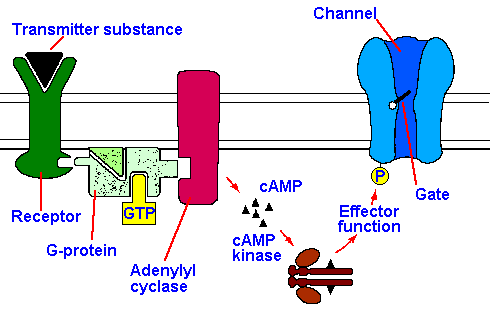 |
Fig. 13-4B. Models of metabotropic synaptic transmission. Here the second messenger is shown as cAMP, but cGMP or another substance or even several different substances could serve as well.
|
The longer latency and
duration of norepinephrine effects
on sympathetic postsynaptic
structures have been explained by
invoking a second messenger, in
this case, cyclic adenosine
monophosphate (cAMP). The
model proposed for this kind of
transmission, called metabotropic
transmission, is shown
schematically in Figure 13-4B.
According to the model, the
transmitter substance interacts with the receptor on the postsynaptic membrane, activating
adenylate cyclase and producing cAMP. The cAMP activates a protein kinase, phosphorylating a
protein. It is the phosphorylation of the protein which produces the change in membrane
potential, either through a change in membrane ionic conductance or through stimulation of an
electrogenic pump. The process is reversed by dephosphorylating the protein and hydrolyzing
cAMP. Often associated with metabotropic transmission is an increase in membrane resistance, not a
decrease as in ionotropic transmission.
It has also been suggested that the longer latencies and durations of the muscarinic effects of
ACh and the effects of dopamine and norepinephrine may be partly the result of having long
diffusion distances between the site of release at the presynaptic terminal and the site of action,
i.e., that the synapses differ from the classical 15- to 200-nm cleft variety. These have sometimes
been called nonsynaptic interactions, but the current trend is to call them loose synapses. Longer
synaptic delays can readily accommodate longer diffusion times. Of course, this usage makes transmitter substances subtly merge with neurohormones.
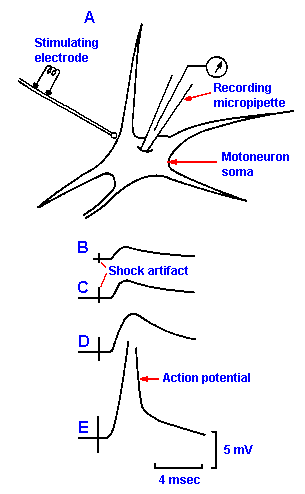 |
Fig. 13-5. A. The method of recording the postsynaptic potential with a micropipette. B-E. EPSPs elicitied in a motoneuron by stimuli to a peripheral nerve at increasing strengths. Note that the critical firing level is achieved and that the action potential is initiated in E. (Eccles JC, Eccles RM, Lundberg A: J Physiol (Lond) 136:527-546, 1957)
|
Table 13-1 shows that glycine, GABA, L-glutamate, and L-aspartate are probably ionotropic
transmitter substances. Norepinephrine, dopamine, serotonin, epinephrine, histamine, and
probably substance P are metabotropic. Acetylcholine is sometimes ionotropic (at nicotinic
cholinergic synapses; those also activated by nicotine), and sometimes metabotropic (at
muscarinic cholinergic synapses; those also activated by muscarine). The second messenger at
metabotropic (muscarinic) cholinergic synapses is thought to be cyclic guanidine monophosphate
(cGMP).
The excitatory postsynaptic potential The postsynaptic cell
can either hyperpolarize or hypopolarize in response to the
transmitter substance. It is possible to study the postsynaptic
events by puncturing the soma of a cell with a microelectrode to
pick up the changes in membrane potential that result from
activity in a presynaptic axon. The arrangement of the cell and
the electrode is shown in Figure 13-5 along with some sample
records. If we stimulate a peripheral nerve at weak intensity
while recording from a motoneuron, we see in the record a
hypopolarization with a latency of 1-2 msec, followed shortly by
a repolarization of the cell. This response is the excitatory postsynaptic potential, or EPSP, shown in Figure 13-5B. It
has a decay time-constant of about four msec and, like the
generator potential, is a local or nonpropagated event. The
EPSP is in fact a generator potential; it is sufficiently important
to warrant special consideration and a name of its own. The
amplitude of the EPSP varies with the strength of the stimulus
(that is, with the number of fibers stimulated), as shown in Figure
13-5C-E, and if it hypopolarizes the membrane to or beyond its
critical firing level, the motoneuron will discharge an action potential (Fig. 13-5E).
The neuromuscular junction: an example of excitatory transmission
We can use transmission at the neuromuscular junction as an example of excitatory
transmission because it displays many of the important features of such transmission. It is also of
historical interest because it was in the neuromuscular junction that most of the early studies of
synaptic transmission were done. It is easily accessible, and the postsynaptic cell (in this case a
striated muscle cell) is large and easily penetrated by recording electrodes. The neuromuscular
junction or motor end-plate is morphologically much like any other synapse except that it is
larger. The axon of the motoneuron loses its myelin sheath near its termination and expands
greatly, forming a bouton that is 10 micrometers or so in diameter. It contains synaptic vesicles, known to
contain the transmitter substance, acetylcholine, in its terminal. The postsynaptic element in this
synapse is a skeletal muscle cell whose membrane is specialized at the end-plate by numerous
deep infoldings. Only the peaks of these folds are near the motoneuron terminal, but the depths of
the folds are in contact with the fluid of the synaptic cleft. The structure of the neuromuscular
junction is shown in Figure 13-6.
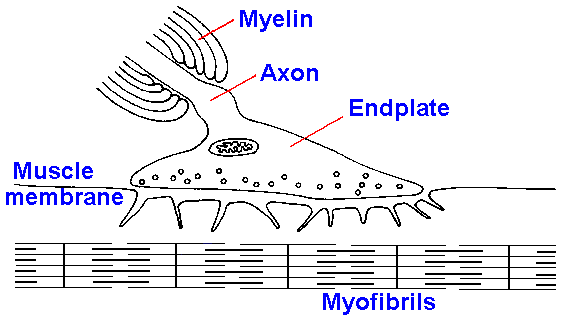 |
Fig. 13-6. Structure of the neuromuscular junction. The terminal of the motoneuron is expanded over the synaptic cleft. Muscle membrane is deeply infolded under the terminal.
|
It is possible to remove the muscle
and its nerve from an organism and put it
into a bath of Ringer's solution (a
balanced ionic solution, the ionic
constituency of which is similar to that of
blood). The nature of such a preparation
is shown in Figure 13-7. When a
microelectrode is used to penetrate the
muscle, a resting membrane potential
(Vr) of about -70 mV is recorded as
described in Chapter 3.
When the axon of the motoneuron is
stimulated, it can be excited to discharge
a spike that conducts down to the
terminal. Hypopolarization of the
terminal by the action potential opens
the voltage-gated Ca++ channels, Ca++
enters the terminal, the synaptic
vesicles fuse with the terminal
membrane, and acetylcholine is
released into the synaptic cleft. The
acetylcholine diffuses across the
synaptic cleft from presynaptic to postsynaptic in less than 100 microseconds and
interacts with the acetylcholine
receptors on the folded postsynaptic,
muscle membrane. The acetylcholine
receptor is believed to be a pentameric glycoprotein composed of 4 types of transmembrane
polypeptides. Two acetylcholine molecules bind to the pentamer with weak cooperativity and
cause a conformational change. The binding of the acetylcholine opens the channel which has an
effective diameter of 0.65 nm, and it is nonselective, i.e., it is permeable to small ions-Ca++, Na+,
K+ and Cl-. Because the channel is nonselective, the contribution that any ion makes to the total
current is a function of its driving force and concentration. As a result the total ionic current is
comprised mainly of Na+ current because of its large electrochemical gradient at the resting
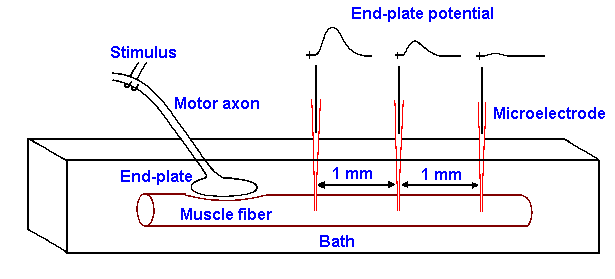 |
Fig. 13-7. Recording setup for the end-plate potential. Muscle is bathed in Ringer's solution containing curare, and motor axon is placed on stimulating electrodes. A recording micropipette penetrates the muscle membrane at 1-mm intervals away from the end-plate. Sample records show the nature of the recording at each site.
|
membrane potential and its high external concentration. Inward Na+ current hypopolarizes the
membrane and, when the membrane potential crosses the critical firing level, an action potential is
initiated in the muscle membrane. In Figure 13-7, the instant when the stimulus is applied to the
nerve is signaled by the stimulus artifact (a nearly instantaneous spread of stimulus current from
stimulating to recording electrodes) and then, after a suitable time for conduction along the axon
and for synaptic delay, the membrane hypopolarizes, leading
to the action potential.
If the preparation has been bathed in a solution containing
the drug curare, a substance used experimentally as a muscle
relaxant and used by South American Indians as a poison that
is applied to arrowheads, the stimulus to the nerve leads to a
change in potential, called the end-plate potential. The end-plate potential looks and behaves much like EPSPs in nerve
cells; an example is shown in Figure 13-8B for comparison
with the uncurarized spike in A(4). Like the EPSP, the end-plate potential is graded and non-propagated, as shown by
the decreasing amplitude as the potential is recorded further
from the end plate (reading traces toward the right in Figure 13-7). As the electrode is moved
farther and farther along the muscle membrane away from the end-plate, the potential drops in
amplitude because it conducts electrotonically. As in other synapses, the receptor region of the
postsynaptic membrane contains no voltage-gated channels and therefore does not discharge
spikes, but the end-plate potential has only to bring the adjacent membrane to critical firing level
to initiate an action potential there. Normally, in the absence of curare, the end-plate potential is
more than large enough to bring the membrane to the critical firing level; an action potential is
initiated in the muscle fiber for each spike in the motoneuron connected to it.
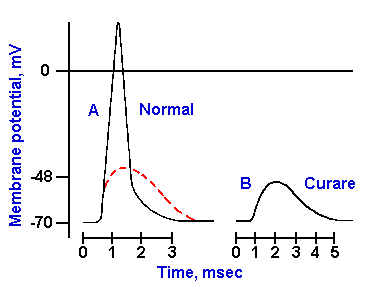 |
Fig. 13-8. The spike and the end-plate potential. A. The spike recorded by a transmembrane micropipette from the muscle. B. The end-plate potential recorded after the spike has been blocked with curare.
|
Acetylcholine acts to open the ionic channels in the postsynaptic membrane for only 1-2 msec;
the remainder of the end-plate potential is simply due to the passive properties of the muscle
membrane. After 1-2 msec, the acetylcholine is removed from the receptor and hydrolyzed by the
enzyme acetylcholinesterase, which is found on the postsynaptic membrane mainly in the region of
the synaptic cleft. Acetylcholinesterase is capable of hydrolyzing about 10 acetylcholine
molecules per msec. Therefore, most of the transmitter substance is hydrolyzed, and the products
are taken up again by the motoneuron terminal, but some of the acetylcholine escapes from the
cleft and is carried away by the blood. There is no danger that this escaped transmitter can re-excite the muscle because it is only effective in changing the membrane potential when applied to
the membrane within the cleft, i.e., there are no active receptors on the muscle membrane except
in the cleft.
Within the motoneuron terminals, acetylcholine is stored in the synaptic vesicles, each vesicle
containing about 10,000 molecules of acetylcholine. Because the transmitter substance is stored
in this fashion and because the entire content of a vesicle is released or none of it is released, it is
reasonable to assume that the end-plate potentials must be made up of some multiple of the
potential change caused by a single vesicle's contents. Actually, when recordings are made from a
nerve-muscle preparation (Fig. 13-7) at rest, small, spontaneous hypopolarizations are seen in the
membrane potential. These have a time-course and configuration reminiscent of the end-plate
potential, and they are termed miniature endplate potentials or MEPPs. Pharmacologically and
physiologically, it has been demonstrated that MEPPs have the same properties as the end-plate
potential and that they are caused by spontaneous release of small quantities of acetylcholine.
MEPPs tend to have the same amplitude or multiples of their smallest amplitude, so it is surmised
that they are caused by release of nearly equal-sized packets of acetylcholine, called quanta. It
seems reasonable to assume that a quantum is the amount of transmitter in a single vesicle. It also
seems reasonable that the end-plate potential is always composed of integral multiples of a MEPP.
MEPPs occur spontaneously at irregular intervals with a low rate of occurrence at the resting
end-plate. The motoneuron spike increases the rate of occurrence of MEPPs for a short time, 1-2
msec, during which 200-2,000 quanta, depending upon the particular nerve-muscle preparation,
are released. Each quantum opens up to 2,000 channels and each channel can admit as many as
20,000 Na+ ions. The potential for change in the membrane potential of the muscle is enormous
and therefore always more than enough to cause a spike in normal, healthy muscle.
Because the process of transmission at the neuromuscular junction contains so many different
steps, the possibilities for interference with the process are numerous. Curare has already been
mentioned as a blocker; curare competes with acetylcholine for the receptor sites, but is incapable
of activating them. Other competitive blockers, such as decamethonium or succinylcholine, not
only compete, but activate the receptor. Their paralyzing effect is due to the tenacity with which
they bind the receptor and the difficulty with which they are removed from the receptor and
hydrolyzed.
The toxin of botulinus bacteria, found in some spoiled food, is an extremely powerful
neuromuscular blocker that works by preventing the release of the transmitter substance from the
motoneuron terminals. Low Ca++ concentrations in the extracellular fluid also prevent release of
transmitter substance. Elevated Mg++ or Mn++ levels also block release, but they work by
competition with Ca++.
Neuromuscular blocks can also be created by interfering with the action of the ACh-degrading
enzyme acetylcholinesterase. Cholinesterase inhibitors, such as neostigmine, block the removal
and hydrolyzation of acetylcholine; thus, the muscle membrane stays hypopolarized for too long a
period and the muscle cannot relax. This is the principle of action of many insecticides and nerve
gases. Prolonged hypopolarizations lead to convulsions, then to paralysis and death, usually
caused by paralysis of the diaphragm.
The disease myasthenia gravis is characterized by muscle weakness after repeated activation
of the neuromuscular junction, but not for a single activation. Patients typically are strong in the
morning, but become progressively weaker as the day goes on. The problem with the myasthenic
appears to be threefold: (1) there is an abnormality of the immune system such that antibodies are
formed against the acetylcholine receptor, and the number of receptors is reduced; (2) there is a
decreased ability to resynthesize acetylcholine from the acetate and choline taken up into the
terminals; and (3) there is increased hydrolysis of acetylcholine. With repeated activation of the
synapse, the vesicular stores of acetylcholine are depleted, not replenished as they normally would
be, and after a while the junction fails to transmit. In addition, the reduction in the number of
receptors means that less of the acetylcholine available can be bound. Administration of
anticholinesterase drugs like neostigmine is sometime effective in treatment of myasthenia,
probably because they make the acetylcholine remain in the cleft longer, increasing the likelihood
that it will bind to a receptor and activate the muscle.
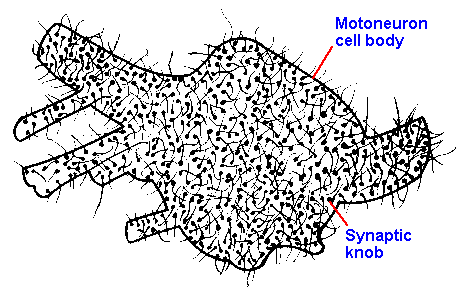 |
Fig. 13-9. The surface of a motoneuron soma studded with boutons terminaux. (Schadé JP, Ford DH: Basic Neurology. Amsterdam, Elsevier, 1965)
|
Synaptic transmission between neurons
Synaptic transmission between neurons is basically
the same as at the neuromuscular junctions.
Transmitter substance release is triggered by terminal
hypopolarization and is dependent upon Ca++. The
release process is quantal in nature, and transmitter
release occurs spontaneously. Synaptic transmission
is terminated by removal of the transmitter substance
from the synaptic cleft through reuptake or
hydrolysis. One fundamental difference is that in
synaptic transmission between neurons, an EPSP in
the postsynaptic neuron, caused by a single spike at a
single synapse, seldom is large enough to trigger a spike. In general, the EPSPs caused by a
single presynaptic action potential are small, only 1-2 mV of hypopolarization, and cannot bring
the postsynaptic cell to critical firing level. For the postsynaptic cell to be excited to discharge
requires spatial and temporal summation, the same two phenomena discussed previously. Figure
13-9 shows a sketch of the soma of a motoneuron, literally encrusted with boutons from afferent
fibers of various sources, to show how extensive neural interconnections are. Estimates of the
density of synapses on motoneurons have indicated that boutons cover 40-50% of the somatic and
50-80% of the dendritic surfaces. If we stimulate more than one of these afferent fibers with a
single stimulus, then each active fiber will release its transmitter substance onto the membrane of
the motoneuron and cause its own EPSP, and we see the algebraic sum of them. In Figure 13-10A are illustrated the EPSPs elicited in a motoneuron by two action potentials in two different
afferent fibers, when stimulated separately and when stimulated together. The response to
simultaneous stimulation is clearly seen to be the algebraic sum of the two single responses. This
is an example of spatial summation.
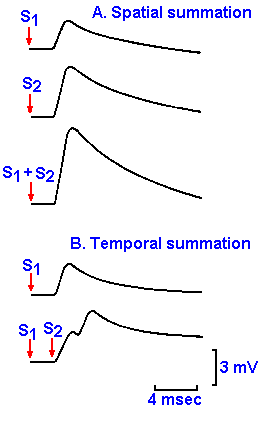 |
Fig. 13-10. Spatial and temporal summation of EPSPs. A. EPSPs elicited by stimulation of two fibers afferent to a motoneuron both separately and simultaneously. Notice the algebraic summation. B. EPSPs elicitied by a single stimulus to a fiber afferent to a motoneuron and two stimuli applied in rapid succession (2 msec apart).
|
Like all generator potentials, EPSPs can show temporal
summation. If the same afferent axon is stimulated twice in rapid
succession, the response to the first stimulus is not yet over before
the response to the second begins. The changes in potential again
sum, but because they do not start at exactly the same time (2 msec
apart), the sum is a bit irregular in shape (Fig. 13-10B). This is an
example of temporal summation. The membrane potential can be
brought to firing level by summation, spatial or temporal or both.
Any number of EPSPs, from a variety of types of presynaptic
neurons, can be summed by a single postsynaptic cell.
Synapses that require a lot of summation to reach firing level are
called integrative synapses, and they make up the bulk of the
synapses in the nervous system. A few synapses require only one
presynaptic action potential to bring the postsynaptic membrane to
critical firing level, and these are called obligatory synapses. An
example of an obligatory synapse is the neuromuscular junction. A
single action potential in a single alpha-motoneuron causes a postsynaptic action potential in every
extrafusal muscle fiber in its motor unit (a motoneuron plus all the muscle fibers it innervates is a
motor unit). Generation of the action potentials in the alpha-motoneurons themselves requires
considerable summation, and therefore synapses on motoneuron somata or dendrites are
integrative. Not all neurons behave exactly like motoneurons, but most use this same basic
mechanism for transmission at chemical synaptic junctions with other cells.
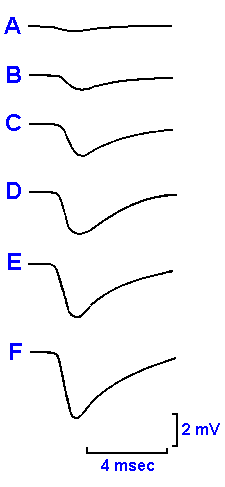 |
Fig. 13-11. An IPSP elicited by a single stimulus applied to a peripheral nerve at increasing strengths from top to bottom.
|
The inhibitory postsynaptic potential Another difference between
neuronal and neuromuscular synapses is the possibility of inhibition at
neuronal synapses. Inhibition is not seen in mammalian neuromuscular
junctions. With an appropriate stimulus, the response of the motoneuron
can be an hyperpolarization, beginning 3-4 msec after the stimulus, followed
by a return to the resting potential, again with a decay time-constant of
about four msec. This response is called the inhibitory postsynaptic
potential, or IPSP, because it drives the membrane 1-4 mV away from the
critical firing level and therefore reduces the frequency or, alternatively, the
probability of firing of the postsynaptic cell. Figure 13-11 shows that as the
stimulus strength increases (from top to bottom), so does the amplitude of
the IPSP. Like the EPSP, the IPSP is a nonpropagated event. IPSPs can
sum either spatially or temporally to hyperpolarize the cell to an even greater
extent than a single IPSP. IPSPs are never seen in mammalian muscles.
Ionic mechanisms of postsynaptic potentials It appears that the
excitatory transmitter substances responsible for the EPSP act at the
postsynaptic membrane by increasing the permeability of the membrane to
all small ions, including sodium, potassium, calcium and chloride ions; there
is an increase in net flux of ions, with all ions moving down their
electrochemical gradients. The major contributor to the change in potential is sodium current
because of the large driving force (membrane potential minus equilibrium potential) on sodium
ions and the large change in sodium conductance. It is also apparent that the change in the
membrane's permeability to sodium is much larger than the change for potassium, because the
potential that the membrane is seeking during the EPSP, the equilibrium potential of the EPSP, is
0 to +30 mV, the exact value depending upon what synapse is being studied. Recall that the
resting membrane's permeability to sodium is only 1/30th of that to potassium, making the resting
potential very near the potassium equilibrium potential. If the membrane is suddenly made 30
times more permeable to sodium ions, the membrane potential will shift to a level halfway
between the sodium and potassium equilibrium potentials, about -15 mV. In fact, during the
EPSP the membrane potential shifts farther, to 0 to +30 mV, so the relative change in sodium
permeability must make the permeability ratio much greater than 1:30, in fact, greater than 1:1.
Apparently, chloride plays no major role in this process, but any chloride current would tend to
hold the membrane near VCl-, i.e., near Vr.
The IPSP, on the other hand, is produced by increased permeability of the membrane to
chloride or potassium or both. If, in the postsynaptic cell, Vr=VCl-, then chloride current will be
zero, and chloride will make no contribution to the hyperpolarization. If Vr is less negative than
VCl-, then the driving force on chloride will move it inward, creating an outward current. Under
this circumstance, an increase in chloride conductance will hyperpolarize the membrane. On the
other hand, if Vr is more negative than VCl-, then the driving force on chloride will move it
outward, creating an inward current. Under this circumstance, an increase in chloride
conductance will actually hypopolarize the cell. Vr is always less negative than VK+, so an
increase in potassium conductance always results in hyperpolarization. Chloride and potassium
are nearly in electrochemical equilibrium, and therefore their driving forces are small, but then the
amplitude of the IPSP is also small.
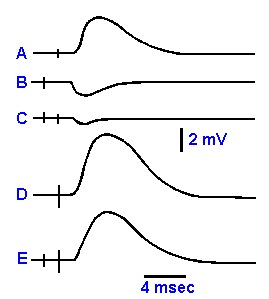 |
Fig. 13-12. The effectiveness of an IPSP in reducing EPSPs. A. The EPSP by itself. B. The IPSP by itself. C. The EPSP and IPSP initiated at the same time. D. A much larger EPSP by itself. E. The EPSP in D with the IPSP in B.
|
It is important that the inhibitory effect of the IPSP is not due
simply to the hyperpolarization, driving the membrane away from
the critical firing level, but also includes another process. If the
effect were simply due to the hyperpolarization, then summation
between EPSPs and IPSPs would still be linear, IPSPs summing
with a negative sign. The maximum change in voltage of the EPSP
is 8-10 mV, whereas that for the IPSP is 2-4 mV; yet an IPSP can
reduce the amplitude of an EPSP by more than its own amplitude,
that is, they do not sum algebraically. This is shown in Figure 13-12. When the appropriate pathways to a cell are stimulated, an
EPSP and an IPSP are initiated in the cell, as shown in A and B. If
the EPSP and the IPSP are initiated at the same time, the resulting
change in potential is a small hyperpolarization as shown in C, not
the small hypopolarization that would be expected if there were algebraic summation. An EPSP
(D) larger than the IPSP (in terms of amplitude) must be added to the response to hypopolarize
the cell to the same level as the original EPSP, as shown in E. This happens because the IPSP
results from an increased membrane conductance for K+ and Cl-. This means that the membrane
resistance (R=1/g) is decreased. The inward Na+ current of the EPSP therefore produces a
smaller voltage drop across the smaller membrane resistance (V = iR, R is reduced) and a smaller EPSP. The
IPSP inhibits both by virtue of the decreased membrane resistance and the hyperpolarization. The
hyperpolarization forces a greater amount of hypopolarization to achieve critical firing level, and
the decreased membrane resistance reduces the size of the EPSP. For this reason, inhibition
always makes itself felt. Equal excitatory and inhibitory presynaptic inputs to a cell always result
in inhibition of its discharge. And it is for this reason that increasing Cl- conductance inhibits even though Cl- is in electrochemical equilibrium and changing it conductance produces no voltage change in the cell.
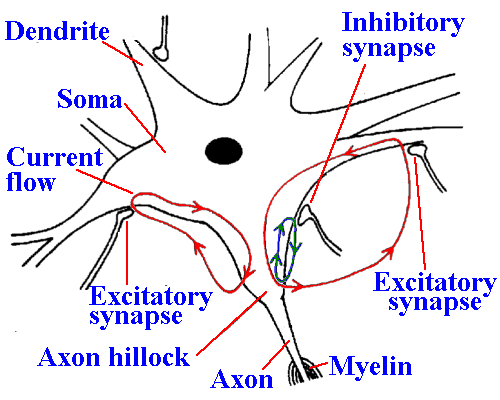 |
Fig. 13-13. Current flow at synapses. Excitatory and inhibitory synapses are indicated on the soma and proximal dendrites and currents initiated at each, flowing through the possynaptic membrane and the region of the axon hillock, where the spike is thought to be initiated.
|
The postsynaptic membranes at all synapses are
electrically inexcitable; the action potential is initiated
somewhere else on the membrane. Most people
think that the spike in most neurons is initiated in the
region where the axon is connected to the soma, the
axon hillock. The axon hillock is thought to have an
electrical threshold about half that of the soma and
dendrites, so that the spike is initiated there first.
Figure 13-13 shows a drawing of the neuron
showing the axon hillock, synaptic junctions on the
soma and dendrites, and the currents that flow when
the synapses are active. EPSPs and IPSPs are not
propagated, but spread electrotonically into the
region of the hillock. This means that, for synapses
producing equal changes in membrane potential at the synapse, the ones closer to the hillock have
a greater influence on the firing of the cell. However, synapses on dendrites tend to generate
particularly large EPSPs, somewhat offsetting their greater distance. Inhibitory synapses,
synapses that produce IPSPs, also tend to be located closer than excitatory synapses to the axon
hillock (the spike-generating region). This arrangement may also add to the great influence of
IPSPs on the neuron membrane.
The firing pattern of the
neuron is completely determined
by the sum of its synaptic
bombardment, both excitatory and
inhibitory. This is especially
important in cells with integrative
synapses. It was pointed out in the
earlier discussion of generator
potentials that there is a linear
relationship between generator
potential amplitude and the
frequency of discharge in the
receptor or its nerve. The EPSP is
a generator potential, and, like any
good generator potential, its amplitude is related to discharge frequency in a linear way. The
hypopolarization at the axon hillock is increased by summation of EPSPs from different synapses
and decreased by summation of IPSPs. As a result, the firing frequency increases or decreases.
Figure 13-14 shows a hypothetical arrangement of synapses on a cell and some different
configurations of synaptic potentials at the axon hillock (A)(5) and the resulting patterns of
discharge in the axon hillock (B). Shown are two excitatory synapses, one on the soma and one
on a dendrite, and one inhibitory synapse on the soma. Each pattern in A was generated by some
combination of inputs over the three synapses (indicated by the letters: a, b and c). The same synapses are
involved in generating each pattern; only the order and timing are changed. Even small changes
result in noticeably different patterns of spike discharges, with great consequences for behavior.
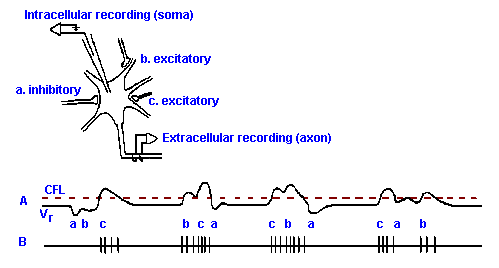 |
Fig. 13-14. The combination of EPSPs and IPSPs to generate different patterns of spike discharge. Inset shows a neuron with three synaptic junctions--two on the soma and one on a proximal dendrite. Two of the synapses are excitatory; one is inhibitory. A. Transmembrane potential recorded with the micropipette, with different temporal arrangements of a single postsynaptic potential at each synapse (indicated by letters under trace). For the purpose of illustration, EPSPs and IPSPs are shown larger than normal; they can cause the membrane potential to cross the critical firing level (CFL). B. The spike trains that would be recorded from the axon of the cell as generated by the synaptic potential patterns in A.
|
If the precise timing required to produce spatial and temporal summation is altered, the
precise firing patterns of interneurons and motoneurons required to produce even the simplest of
movements are no longer possible. In fact, the real impact of certain demyelinating diseases such
as multiple sclerosis is not due to destruction of neurons or even blockage of conduction; they still
conduct (at least in the early stages of the disease), although at reduced velocities after the myelin
is removed. The slowed conduction in a demyelinating disease means that some impulses do not
arrive on time at synapses on motoneurons. Considering the time constant of an EPSP, a slowing
of conduction that produces even a 0.5-msec delay in the arrival of an impulse at the synapse can
have devastating effects on movement. The timing of the arrival of impulses is also important in
sensory events. Humans use small differences in the time of arrival of a sound at the two ears to
localize the source of sounds with low frequencies. This difference in arrival time can be as little as 30 sec. Clearly, very close timing of impulses is essential to this behavior.
Rectification(6) In the initial discussion of the initiation of an action potential in axons, it was
noted that, with electrical stimulation, once the critical firing level is reached, the action potential
propagates in both directions away from the point of stimulation. The direction normally taken by
action potentials is the orthodromic direction; the reverse is the antidromic direction. If the
action potentials can travel down an axon both ways, why is there a "normal" direction? The
answer lies in the synapse. When the action potential reaches a synapse it can go no further, but it
can cause a release of transmitter substance in a presynaptic element. There is usually no transmitter
mechanism in the postsynaptic element; therefore, synapses act as rectifiers, allowing transmission
in only one direction.
Modulatory role of transmitter substances The term neuromodulator has been coined to
describe certain functions of transmitter substances (or putative transmitter substances). A
transmitter substance acts as a neuromodulator when it alters the synaptic action of other neural
inputs by means other than itself producing direct excitation or inhibition. In other words, it acts
by means other than eliciting EPSPs or IPSPs. Neuromodulators can change the release of a
transmitter substance from presynaptic terminals. This can be accomplished by way of
autoreceptors, which when bound by a transmitter substance, modulate further release of that
substance, or it can occur when one transmitter substance modulates the release of another.
Norepinephrine at some synapses in the autonomic nervous system can inhibit its further release.
When enkephalin is released into sympathetic ganglia by preganglionic neural input, it can inhibit
the release of acetylcholine within that ganglion.
In some cases, the postsynaptic potential elicited by a given transmitter substance can be
altered by, or contingent upon, the postsynaptic action of a neuromodulator. For example, a brief
exposure to dopamine released synaptically into sympathetic ganglia enhances the muscarinic
hypopolarizations induced by acetylcholine for hours, even though the dopamine causes no
change in the membrane potential or resistance of the postsynaptic cell. Similar effects of
dopamine have also been described in the caudate nucleus and hippocampus. A shorter
potentiation of both excitatory and inhibitory responses of Purkinje cells in the cerebellar cortex is
induced by norepinephrine released by axons originating in the locus ceruleus.
It has been suggested that these longer lasting changes in neural activity produced by
neuromodulators may play a role in slowly developing and enduring behavioral changes such as
learning and memory. The effects of muscarinic antagonist drugs on learning and monoamines on
sleep/waking and learning may indicate that this suggestion has some credence.
| Physiology of an electrotonic synapse |
In known examples of electrotonic synapses in invertebrates, the anatomical substrate of
transmission is a gap junction in which the joined membrane is of lower resistance than
surrounding membrane, i.e., the cell-to-cell resistance is less than the cell-to-extracellular fluid
resistance, and it is electrically inexcitable, i.e., it does not generate action potentials. An impulse
propagates into the region of the junction, and the resulting current flows across the junctional
membrane, out of the presynaptic cell and into the postsynaptic cell. That current must again flow
out of the postsynaptic cell (according to Kirchhoff's current law), this time through electrically
excitable membrane, causing an hypopolarization, which, if large enough, can cause the
postsynaptic cell to discharge. Usually, though, the resulting hypopolarization is small, of the
order of 1-2 mV.
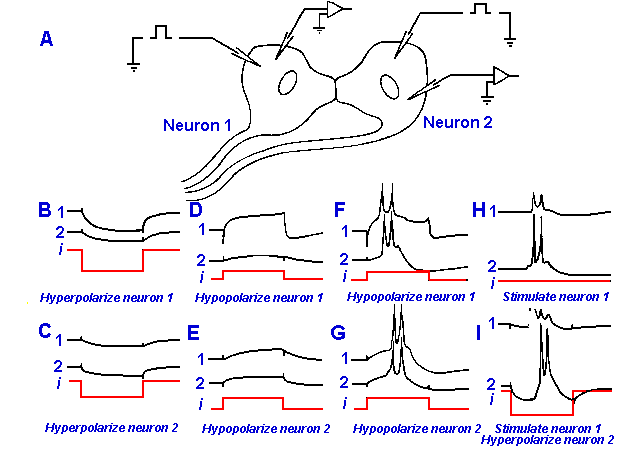 |
Fig. 13-15. Electrotonic spread between spinal neurons in an electric fish. A. Recordings are made simultaneously of the membrane potentials of two neurons while either one is hypopolarized or hyperpolarized through an intracellular electrode. B-G. The responses of both cells are shown to hyperpolarization of neuron 1 (B) or neuron 2 (C), hypopolarization of neuron 1 below (D) and exceeding its critical firing level (F), and hypopolarization of neuron 2 below (E) and exceeding its critical firing level (G). H. An impulse is initiated by a brief stimulus to neuron 1, and it spreads to neuron 2. I. The same as in H except that neuron 2 is strongly hyperpolarized; this fails to block spread of the spike from neuron 1 to neuron 2. (Bennett MVL, Pappas GD, Aljure E et al.: J Neurophysiol 30:180-208, 1967)
|
Figure 13-15 shows recordings
made intracellularly from two
spinal motoneurons that drive the
electric organ of the electric fish,
Gnathonemus. The electric organ
emits electrical impulses of 0.3-msec duration at frequencies of up
to 100/sec and presumably is used
by the fish for guidance or perhaps
communication. The recording
arrangement is shown in A.
Histologically, these motoneurons
have been shown to be
interconnected by thick dendritic
or somatic processes, as shown.
The recordings in B-I are made
from neurons 1 (marked 1), from
neuron 2 (marked 2), and from the
current applied to the cells
(marked i). An hyperpolarizing current was applied to neuron 1, and responses were recorded
from both neurons in Figure 13-15B. Both cells were hyperpolarized. A similar result occurred
when an hyperpolarizing current was applied to neuron 2 (Fig. 13-15C). An hypopolarizing
current, applied to either cell, hypopolarized both or, in some cases, caused both to discharge
action potentials (Fig. 13-15D-G). Notice that the latency of the discharge was longer in the cell
that did not receive the current injection directly. This is due to the slower rate of rise of the
electrotonic potential farther from the current source, causing a delay in reaching the critical firing
level (Fig. 13-15F,G). The sort of reciprocal relationship shown in Figure 13-15 is just what one would expect for such an electrotonic synapse. Current
flows as easily from neuron 1 to neuron 2 as from neuron 2 to neuron 1, i.e., the junction is not
rectifying. There are a few examples in invertebrates of electrotonic synapses that do rectify.
This can be the result if the two cells have greatly different membrane resistances, membrane
areas, or voltage thresholds, the latter being the voltage at which the synapse begins to transmit.
| Chemical and electrotonic synapses compared and contrasted |
Inhibition It should be clear that chemical synapses can produce either hypopolarizations (excitation) or hyperpolarizations (inhibition) in the postsynaptic cell. As far as we know, axons
do not carry propagated hyperpolarizations in any nervous system, and therefore simple hyperpolarizing postsynaptic potentials will not occur at electrical synapses. There is the possibility of
transmission of hyperpolarizing after-potentials at electrical synapses, especially if transmission at
the junction were rectified in the proper direction. If the polarity of rectification is such that
hypopolarizations pass more easily from pre- to postsynaptic and hyperpolarizations from post- to
presynaptic, a large hyperpolarizing after-potential in the postsynaptic cell would then produce a feedback inhibition of the presynaptic
cell with little feedback excitation (Table 13-3). Such a possibility has been suggested for
synapses in Aplysia.
Table 13-3
Properties of Single Chemical and Electrical Synapses
|
| Property |
Chemical synapses |
Electrical synapses |
| 1. Rectification |
Always |
Sometimes, usually not |
| 2. Amplification |
Yes |
No |
| 3. Delay |
Yes |
No |
| 4. Inhibition |
Yes |
Yes |
| 5. Summation |
Yes |
Yes, but over shorter time |
| 6. Influenced by membrane
potential |
Yes |
No |
The advantages of chemically mediated inhibition are that it can be larger in size, i.e., a greater
hyperpolarization; longer in duration; and not limited in its site of application. Electrically
mediated inhibition would occur more rapidly, because of the lack of synaptic delay, but this need
not be a big advantage. The CNS could easily produce small time compensations.
Synaptic delay Transmission at a chemical synapse requires mobilization of synaptic vesicles,
exocytosis, diffusion of a transmitter substance (in some cases over long distances), reaction of
the transmitter substance with postsynaptic receptor sites, production of changes in membrane
permeability, and a change in membrane potential, produced either directly or through a second
messenger. All of these steps take time. This time is called the synaptic delay, and it is
measured as the time between arrival of the impulse at the presynaptic terminal and the start of the
postsynaptic response. In mammals, the ionotropic synaptic delay is of the order of 0.1-0.3 msec.
At the neuromuscular junction, most of the delay consists of the time required for release of the
chemical transmitter substance. Diffusion time and onset of permeability changes apparently
contribute little to it.
The process of electrical transmission occurs with minimal delay, usually less than 0.05 msec.
It is this short delay that gives electrical synapses their usefulness as neural synchronizers. The
electrical interconnection of neurons causes them to tend to fire synchronously. This is
presumably of advantage when rapid movements or high-frequency events, such as electric organ
discharges, are being controlled. What other advantages it may confer on a system are not
known.
Rectification Because of the locations of synaptic vesicles and receptors for transmitter
substance at chemical synapses, the latter are, of necessity, rectifying junctions, i.e., they allow
transmission in only one direction, as we have already discussed. In cases of reciprocal synapses,
there is a mechanism for removing rectification, but it is not known how pervasive this mechanism
may be. It does exist in the retina and the olfactory bulb. As we have already mentioned, most
electrical synapses appear to be nonrectifying; however, the study of rectification requires
intracellular recording and stimulation of both the pre- and postsynaptic elements of the synapse,
which is seldom possible in vertebrates.
Summation The processes of spatial and temporal summation in chemical synapses are
general integrative properties of neurons and fundamental to operation of the nervous system.
They are also properties of electrical synapses. The major difference between summation by
chemical and by electrical mechanisms is the longer time-course of chemical summation. The postsynaptic potentials at chemical synapses typically last 10 msec or longer, whereas those at
electrical synapses seldom outlast the duration of the presynaptic spike, i.e., about 1 msec.
Therefore, summation can occur over a period 10 times (or more) longer at chemical synapses.
Amplification At chemical synapses, there can be an effective amplification of the transmitted
signal such that the electrical energy of the postsynaptic response is greater than that of the
presynaptic response. This can occur if the resistance of the postsynaptic membrane is higher than
that of the presynaptic membrane or if the change in membrane permeability, brought about by a
single presynaptic spike, is extremely large. This may manifest itself as an increase in the number
of action potentials discharged by the postsynaptic cell. Amplification has been observed for
neurons in the dorsal horn of the spinal cord that are connected to cutaneous primary afferent
neurons (Tapper DN, Mann MD: Brain Res 11:688-690, 1968). It probably also occurs at the
synaptic junctions between receptors and other cells that do not generate action potentials
themselves and neurons that do generate action potentials, e.g., bipolar and ganglion cells in the
retina. No such amplification has ever been seen in an electrical synapse.
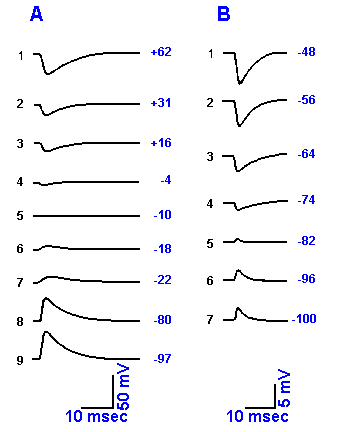 |
Fig. 13-16. Demonstration of the equilibrium potentials of the EPSP and IPSP. A. The influence of changes in membrane potential (indicated at the end of each trace), induced by passing current through an intracellular electrode, on the EPSP recorded from a frog sympathetic gangion cell is shown. The resting potential for this cell was -80 mV, and the equilibrium potential for the EPSP (A5) was -10 mV. (Nishi S, Koketsu K: J Cell Comp Physiol 55:15-30, 1960) B. The influence of membrane potential on the IPSP recorded from a motoneuron in the cat. The resting membrane potential for this cell was -74 mV, and the equilibrium potential for the IPSP was -81 mV (not shown). (Coombs JS, Eccles JC, Fatt P: J Physiol (Lond) 130:326-373, 1955)
|
Influence of membrane potential Because the postsynaptic
potentials at chemical synapses result from changes in
permeability of the postsynaptic membrane to sodium, potassium,
chloride, or other ions, their amplitude and polarity are greatly
affected by the polarity and magnitude of the membrane
potential. An example of the influence of membrane potential
upon both EPSPs, these recorded from a frog sympathetic cell,
and IPSPs, recorded from a cat motoneuron, is shown in Figure
13-16. In Figure 13-16, A8 and B4 show the normal
configurations of the EPSP and IPSP, when initiated with the cell
at resting membrane potential. If the cell is hypopolarized, the
IPSP gets larger (Fig. 13-16, B4-1) because the driving forces
get larger as the membrane potential gets further removed from
the equilibrium potentials of the ions whose permeability is
changed, i.e., chloride and potassium. The EPSP, on the other
hand, gets smaller as the membrane is hypopolarized (Fig. 13-16,
A8-6), until it disappears at about -10 mV (Fig. 13-16, A5) and is replaced by a negative-going
potential (Fig. 13-16, A4). The value of the membrane potential at which the EPSP disappears is
called the equilibrium potential for the EPSP, VEPSP, and it is determined by the equilibrium
potentials for the ions whose conductances change during the EPSP, i.e., primarily Na+ and K+,
with a weighting factor related to the amount of conductance change, as indicated in
equation 5 of Chapter 3. If the permeabilities for both Na+ and K+ change, then the equilibrium potential for
the EPSP is somewhere between the sodium equilibrium potential, VNa+, and the potassium
equilibrium potential, VK+. The squid giant synapse has a reversal potential of +20 mV, the cat
motoneuron, 0 mV, and the neuromuscular junction, -15 mV, indicating that different relative
permeability changes for Na+ and K+ ions occur at different synapses. The relative change in
potassium conductance is larger for the neuromuscular junction than for either the cat
motoneuron or squid giant synapse. With further hypopolarization, a new driving force develops,
but this time in the opposite direction, moving the membrane back toward VEPSP (Fig. 13-16, A4-1).
One can see that the IPSP decreases in amplitude and reverses (in this case, at about -81 mV)
to a hypopolarizing potential as the membrane is hyperpolarized (Fig. 13-16, B4-7), whereas the
EPSP gets larger (Fig. 13-16, A5-7). The IPSP gets smaller either because iCl- and iK+ exactly
balance each other (if both ions are involved) or because ix = Vm - Vx = 0 (if only one ion, x, is
involved). At the equilibrium potential for the IPSP, the amplitude of the IPSP is zero because
there is no net membrane current. As the equilibrium potential is exceeded (Fig. 13-16, B5-7), a
net current (two ions) or new driving force (one ion) develops, but it is in the opposite direction,
so the polarity of the IPSP reverses. The EPSP gets larger with membrane hyperpolarization,
primarily because the driving force on Na+ increases as the membrane potential moves further
from the equilibrium potential of the EPSP.
The postsynaptic potential at an electrical synapse is due to a flow of current through the
membrane resistance with no concomitant change in membrane permeability, although an EPSP
may lead to a permeability change in adjacent, electrically excitable membrane. Because the electrical EPSP
is a simple ohmic voltage change (iR drop), it is not influenced by membrane potential, and
therefore it has nearly the same size at any membrane potential. This is shown in Figure 13-15, H
and I. (Of course, if the cell is near critical firing level, the EPSP may have a disproportionate
effect, i.e., the cell may discharge.)
Inhibition of impulse discharge mediated by IPSPs
is called postsynaptic inhibition, because the effect is
exerted directly on the postsynaptic cell. Postsynaptic
inhibition reduces the cell's excitability to all synaptic
inputs. Another mechanism for producing inhibition,
called presynaptic inhibition, involves effects
exerted on a presynaptic axon terminal.
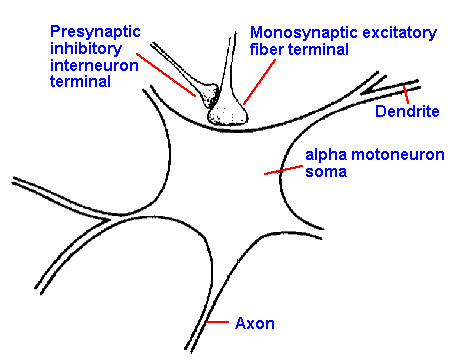 |
Fig. 13-17. The anatomic arrangement of an axoaxonic synapse showing the pre- and postsynaptic axons. Also shown is the axosomatic synapse of a group Ia afferent fiber on the motoneuron. (Eccles JC: The Understanding of the Brain. New York, McGraw-Hill, 1973)
|
To illustrate this inhibition, let us examine the
primary afferent fibers of group Ia. These enter the
spinal cord and go, among other places, to the ventral
(anterior) horn, where they make synaptic contacts on alpha motoneurons that innervate the muscle from which the afferent fibers originated. (This, as we
shall see in Chapter 15, is the anatomical basis of the monosynaptic, tendon tap reflex.) On the
boutons of the Ia afferent fibers there are synapses, i.e., synaptic terminals on synaptic terminals.
The presynaptic terminals on the group Ia terminals come from interneurons that are driven by
group Ia afferent fibers from another muscle. This arrangement is shown in Figure 13-17. The
synapse between the two axons, the axoaxonic synapse, is excitatory in that the presynaptic
(interneuronal, in this case) action potential produces an EPSP in the terminal of the group Ia
afferent fiber.
If the group Ia afferent fiber terminal membrane
is maintained in a slightly hypopolarized condition
and then stimulated to initiate an action potential, the
action potential will be smaller than one that is
initiated when the membrane is at its resting
potential, Vr. This result is illustrated in Figure 13-18(7). Trace B shows the action potential initiated at
Vr = -65 mV. The smaller spikes in C and D were
obtained when the membrane started at hypopolarized values, Vm = -55 mV and -50 mV. The
trace in A shows a larger than normal spike initiated when Vm = -70 mV, i.e., the membrane was
hyperpolarized. (Actually, these records were made in a motoneuron, but the same thing would
presumably happen in the axon terminal.) The spike in C and D is reduced partly because it arises
from a more positive level of membrane potential and partly because the hypopolarization
(previous to the spike) increased the K+ conductance and partially inactivated the Na+
conductance (accommo-dation) in the membrane. The increased K+ conductance and Na+
conductance inactivation reduced the ratio of conductances and therefore the positive overshoot
potential as predictable from equation 5 of Chapter 3. The spike is, therefore, smaller both
because it starts closer to zero and because it does not overshoot as far. This phenomenon is the
basis for the presynaptic inhibitory action. Activity at the axoaxonic synapse partially
hypopolarizes the terminal so that, when an action potential comes down the Ia afferent fiber into
that terminal, its amplitude is reduced. Because the amount of transmitter substance released by a
bouton is proportional to the amplitude of the action potential in it, less transmitter substance is
released, resulting in a smaller EPSP and less excitation of the postsynaptic cell, in this case the
motoneuron. Some measurements indicate that a 15-mV reduction in the amplitude of the
presynaptic spike will reduce the amount of transmitter substance released to 1/10 of its original
value.
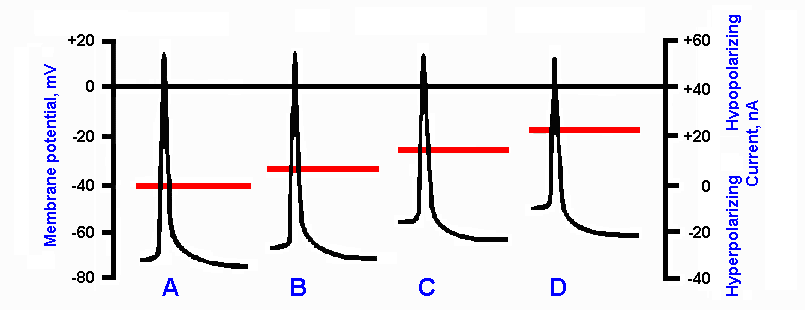 |
Fig. 13-18. Effect of membrane polarization level on the action potential. B. Action potential initiated with membrane at resting potential. A. Spike configuration when cell was previously hyperpolarized by 10 mV. When spike is initated in a hypopolarized membrane, the spike is reduced in amplitude. Examples are shown for 10 mV (C) and 20 mV (D) hypopolarization. Horizontal line through each record indicates the current passed through the membrane to change membrane potential before spike was initiated; values on right ordinate. Membrane potential is indicated on left ordinate. (Eccles JC: The Physiology of Nerve Cells. Baltimore, Johns Hopkins Press, 1968)
|
It is usually said that presynaptic inhibition has no direct effect on the postsynaptic cell, but
this is not entirely true. The EPSP initiated in the Ia bouton causes the release of a small amount
of transmitter substance that raises the level of excitability of the motoneuron slightly. It is
possible that this slight increase in excitability may offset a small amount of the decrease in
transmitter output caused by the spike in the group Ia afferent fiber. In any case, the increased
excitability of the motoneuron will add to the excitatory synaptic activity from other uninhibited
boutons on the same cell. This could give the nervous system a rather subtle way of modulating
excitability in certain pathways, a possible function of this "presynaptic inhibitory circuit" that has
received little attention.
Pre- and postsynaptic inhibition not only differ in their mechanisms but also in their
consequences for the system. With postsynaptic inhibition, the postsynaptic cell is silenced or at
least reduced in its excitability to all inputs no matter what their source. The consequence for the
muscle is that it relaxes because there is no alternate pathway to it. With presynaptic inhibition,
the excitation of the postsynaptic cell (in this case, the alpha-motoneuron) through one synapse is
reduced, but all other synapses perform normally or perhaps even supranormally (see previous
paragraph). For the motoneuron, this means that transmission through group Ia afferent fibers is
reduced, but transmission through polysynaptic and supraspinal pathways is still possible.
All types of primary afferent fibers receive presynaptic inhibition from one source or another.
The terminals of pyramidal tract fibers in the brain stem have been shown to be hypopolarized in
the same manner. The significance of the latter effect is unknown. Presynaptic inhibition may be
found to be more wide-spread as information accumulates in the future. Postsynaptic inhibition
has been found in every structure of the central nervous system, and our ideas of its importance
and ubiquity seem to be increasing. The function of presynaptic inhibition on primary afferent
neurons may be to modulate sensory inputs to the spinal cord at the earliest possible point, before
they can influence spinal cord activity. In this way, the central nervous system can eliminate
unwanted sensory information. By reducing some sensory inputs, presynaptic inhibition may be
useful in certain kinds of contrast enhancement.
| Collateral or recurrent inhibition |
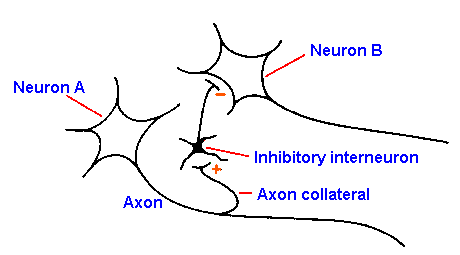 |
Fig. 13-19. A collateral inhibitory circuit. Spike initiated in neuron A invades the collateral to excite the inhibitory interneuron that produces IPSPs in neuron B.
|
A special case of postsynaptic inhibition is
recurrent inhibition, the circuit for which is
shown in Figure 13-19. The output of a neuron,
neuron A, conducts along its axon and out one
axon collateral to excite an interneuron that
inhibits another neuron of the same type as neuron
A, in this case neuron B. Many people draw this
circuit showing that the inhibitory interneuron
inhibits neuron A, making neuron A inhibit itself.
Although this is possible, there is no evidence that
it occurs. The evidence does suggest that neurons
inhibit other neurons of the same type. Thus, neuron A inhibits neuron B and, similarly, neuron B
inhibits neuron A. Thus, the inhibition may more appropriately be termed collateral inhibition.
This is a type of feedback inhibition, in which the output of a neuron is used to inhibit at an
earlier point in the pathway.
Collateral inhibition has been found in the spinal cord and in nearly every major nucleus in the central nervous
system, notably in the cuneate and gracile nuclei, thalamic nuclei, cerebellar nuclei, cerebellar
cortex, and cerebral cortex. One theory suggests that collateral inhibition in the thalamus may
play a role in synchronizing thalamic activity to produce the cortical alpha rhythm (see Chapter
20). It is not known whether this theory can stand empirical tests nor what this kind of inhibition
might be doing elsewhere.
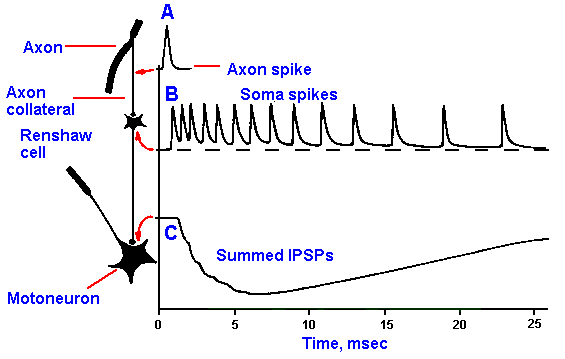 |
Fig. 13-20. Circuit for Renshaw inhibition. Spike (A) generated in the axon at the top of the figure invades the axon collateral, exciting the Renshaw cell. The Renshaw cell discharges a train of spikes (B) in response to a single presynaptic spike (an example of synaptic amplification). The train of spikes in the Renshaw cell produces summed IPSPs in the motoneuron (C). (Eccles JC: The Physiology of Nerve Cells. Baltimore, Johns Hopkins press, 1968)
|
A special case of collateral inhibition is
Renshaw inhibition. This form of
inhibition involves the same circuit as in
Figure 13-19, in which neurons A and B
are alpha-motoneurons, whose axons
innervate skeletal muscles. The Renshaw
circuit is shown in Figure 13-20. The
axon collateral that activates the inhibitory
interneuron is short, remaining within the
ventral horn of the spinal cord and
releasing acetylcholine at its terminal.
(Acetylcholine is also released at the
muscle.) The inhibitory interneuron,
called a Renshaw cell in honor of its
discoverer Birdsey Renshaw, discharges a
burst of spikes (B) in response to a single motoneuron spike (A), releasing a transmitter
substance, perhaps glycine, at terminals on other alpha-motoneurons. This leads to a large, summed
IPSP in these motoneurons (C). Again, there is no evidence that an alpha-motoneuron inhibits itself
through Renshaw inhibition. It has been speculated that Renshaw inhibition may serve to limit
motoneuron firing rates. It is unlikely that this is an important control mechanism for normal
motor activity; motoneurons do not discharge at high rates anyway and, at least during walking,
this kind of inhibition is suppressed. It is possible, however, that this kind of inhibition may play
an important role in preventing or limiting certain kinds of pathological or seizure discharges.
What else it might be doing for an organism is not known.
Synapses are functional connections between cells. Transmission from one cell to another at
most electrical synapses is just like transmission along the membrane of one of the cells.
Transmission at a chemical synapse involves release of a transmitter substance from the
presynaptic element by hypopolarizing its membrane (usually by an action potential). The
transmitter substance can cause an increase in the permeability of the postsynaptic membrane to
small ions, resulting in an EPSP, that is an hypopolarizing potential with a decay time constant of
about four msec and an amplitude of 1-10 mV. The transmitter substance (a different substance
usually) can also cause an increase in permeability to chloride and to potassium ions, resulting in
an IPSP that is an hyperpolarizing potential with a decay time constant of about four msec and an
amplitude of 1-4 mV. Neuromodulators are transmitter substances which can alter the
effectiveness of other transmitter substances in changing the membrane potential of a cell, without
themselves producing any EPSP or IPSP in the cell. EPSPs drive the membrane potential toward
the critical firing level, exciting, and IPSPs drive the membrane potential away from the firing
level, inhibiting. Postsynaptic potentials exhibit spatial and temporal summation. EPSPs do not
add algebraically to IPSPs, because the IPSP's increase in conductance reduces the
hypopolarization produced by the EPSP's ionic current; the IPSP normally predominates.
Inhibition by IPSPs is postsynaptic inhibition. Presynaptic inhibition involves an hypopolarization
of the presynaptic element at a synapse, reducing the spike amplitude in it and thus the amount of
transmitter released by the spike. Presynaptic inhibition reduces transmission through one
pathway afferent to a cell, but not alternative pathways. Postsynaptic inhibition reduces the
excitability of the cell itself and thus the effectiveness of all pathways afferent to the cell. Both
chemical and electrical synapses are capable of rectification, inhibition, and summation. Chemical
synapses are capable of amplification, they have a finite synaptic delay, and they are influenced by
changes in postsynaptic membrane potential. Electrical synapses have not been observed to
amplify input signals. They have essentially no synaptic delay, and they are not influenced by
changing the membrane potential of the postsynaptic cell. Recurrent or collateral inhibition
involves the use of the output of a neuron to inhibit, through an interneuron, other neurons of the same type. Renshaw inhibition is
a special case of recurrent inhibition involving the output of alpha motoneurons and inhibition by
Renshaw cells.
- Bennett MVL: Similarities between chemically and electrically mediated transmission. In
Carlson FD [ed]: Physiological and Biochemical Aspects of Nervous Integration. Englewood
Cliffs NJ, Prentice-Hall, 1968.
- Curtis DR, Johnston GAR: Amino acid transmitters in the mammalian central nervous
system. Ergebn Physiol 69:97-188, 1974.
- Horcholle-Bossavit G: Transmission electrotonique dans le systeme nerveux central des
mammiferes. J Physiol (Paris) 74:349-363, 1978.
- Krnjevic K: Chemical nature of synaptic transmission in vertebrates. Physiol Rev 54:418-540, 1974.
- Libet B: Nonclassical synaptic functions of transmitters. Fed Proceed 45:2678-2686.
- McGeer RL, Eccles JC, McGeer EG: Molecular Neurobiology of the Mammalian Brain.
New York, Plenum Press, 1978.
- Pappas GD, Waxman SG: Synaptic fine structure-morphological correlates of chemical and
electrical transmission. In Pappas GD, Purpura DP [ed]: Structure and Function of Synapses.
New York, Raven Press, 1972.
- Schmidt RF: Presynaptic inhibition in the vertebrate central nervous system. Ergebn Physiol
63:19-101, 1971.
- Tapper DN, Mann MD: Single presynaptic impulse evokes postsynaptic discharge. Brain Res
11:688-690, 1968.
Footnotes:
1. How rapidly the transmitter substance must be hydrolyzed or removed from the synaptic is
determined by the required duration of action of the natural transmitter substance. As we shall see, some
transmitter substances have only short actions; others act for long times; some do both at
different places.
2. Some care should be exercised in attributing single actions to transmitter substances.
Acetylcholine, for example, is usually thought of as excitatory, but there are some cases in which
it is known to be inhibitory.
3. A term used here in the sense of a molecule or molecules that bind specifically the transmitter
substance.
4. Recordings in this figure were obtained using the setup in Fig. 13-7.
5. The spikes have been omitted from trace A for clarity. Obviously, they would be
superimposed on top of the traces in an actual recording. Also the postsynaptic potentials are
shown larger than normal for illustration.
6. Here the term rectification is used in the engineering sense of a lower resistance to
transmission in one direction than in the opposite direction.
7. The horizontal line through each spike indicates the amount of current passed through the
membrane to change the membrane potential before the spike was initiated. Calibrate against the
right ordinate.
[TOC] [Chapter 14] [Glossary] [Index] [Abbreviations]
|